Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| SL Drugs and Pharmaceuticals Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.sldrugs.com | |||
 | +91 (40) 6661-1133 | |||
 | +91 (40) 2375-1130 | |||
 | enquiry@sldrugs.com | |||
| Chemical distributor since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Changzhou Jiuwu Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jiuwuchem.com | |||
 | +86 (519) 8512-1086 +86 13775162768 | |||
 | +86 (519) 8512-5369 | |||
 | info@jiuwuchem.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Matrix Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.matrixscientific.com | |||
 | +1 (803) 788-9494 | |||
 | +1 (803) 788-9419 | |||
 | sales@matrixscientific.com | |||
| Chemical manufacturer | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Carboxylic acid |
|---|---|
| Name | Propoxyacetic acid |
| Synonyms | 2-propoxyacetic acid |
| Molecular Structure | 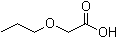 |
| Molecular Formula | C5H10O3 |
| Molecular Weight | 118.13 |
| CAS Registry Number | 54497-00-6 |
| EC Number | 837-785-1 |
| SMILES | CCCOCC(=O)O |
| Solubility | Freely soluble (128 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.054±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Boiling point | 112-115 °C (15 Torr)** |
| Refractive index | 1.40511 (20 °C)*** |
| Flash point | 94.7±13.3 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2013 ACD/Labs) |
| ** | Chen, Li-Tien |
| *** | Palomaa, M. H. |
| Hazard Symbols |   GHS06;GHS07 Danger Details GHS06;GHS07 Danger Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H314-H335 Details | ||||||||||||
| Safety Statements | P260-P261-P264-P271-P280-P301+P330+P331-P302+P361+P354-P304+P340-P305+P354+P338-P316-P319-P321-P363-P403+P233-P405-P501 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
Propoxyacetic acid (C5H8O3) is an organic compound belonging to the class of acetic acids. It consists of a propoxy group (C3H7O) attached to the carboxyl group (-COOH) of acetic acid. The structure of propoxyacetic acid allows it to exhibit both ester-like properties and the reactivity of a carboxylic acid, making it a versatile compound in various chemical and industrial applications. Its discovery is rooted in the study of carboxylic acid derivatives, which are crucial in both biological systems and industrial chemistry. The discovery of propoxyacetic acid can be traced back to the broader exploration of alkyl-substituted acetic acids in the early 20th century. These compounds were synthesized and studied due to their potential in a variety of chemical reactions, such as esterification, amidation, and coupling reactions. Propoxyacetic acid, with its unique combination of an ester-like propoxy group and a carboxylic acid, emerged as an interesting intermediate in organic synthesis. Over time, its applications in the production of herbicides, pharmaceuticals, and other specialty chemicals became more recognized, offering both commercial and scientific value. One of the primary applications of propoxyacetic acid is in the agrochemical industry. It is used as a key intermediate in the synthesis of herbicides and other plant protection products. The compound's structure allows for the attachment of various functional groups that enhance its biological activity. For example, propoxyacetic acid derivatives are often designed to target specific enzymes or metabolic pathways in plants, inhibiting growth or promoting the degradation of harmful compounds. These herbicides are crucial in modern agriculture, helping to control weed populations and increase crop yields. The selective toxicity of propoxyacetic acid derivatives makes them a useful tool in integrated pest management systems. In addition to its use in herbicides, propoxyacetic acid is also utilized in the pharmaceutical industry. The compound serves as an intermediate in the synthesis of bioactive molecules, particularly in the development of drugs that target inflammation, pain, and other conditions. Its carboxylic acid group allows for easy modification and incorporation into various molecular frameworks, while the propoxy group can influence the compound’s lipophilicity and pharmacokinetics. Through these properties, propoxyacetic acid contributes to the development of therapeutic agents with improved potency, selectivity, and bioavailability. Propoxyacetic acid has also found applications in the manufacturing of specialty chemicals and materials. It is used as a building block in the production of resins, plasticizers, and other polymers. These materials are crucial in the production of various consumer goods, including packaging, coatings, and adhesives. The incorporation of propoxyacetic acid into polymer systems can enhance their flexibility, stability, and chemical resistance. Its ability to modify the physical properties of polymers makes it a valuable compound in the field of material science. Furthermore, propoxyacetic acid has been explored for its potential role in environmental chemistry. It is being studied for use in the development of biodegradable materials and green chemistry processes. The compound's structure, which includes both an ester and an acid group, enables it to participate in environmentally friendly reactions, such as those that lead to the breakdown of pollutants or the creation of sustainable products. In summary, propoxyacetic acid is an important compound with a wide range of applications across agrochemicals, pharmaceuticals, materials science, and environmental chemistry. Its discovery as an alkyl-substituted acetic acid derivative has opened the door to various industrial processes and research opportunities. With its ability to influence the properties of other compounds, propoxyacetic acid continues to be a valuable building block in the development of new materials, chemicals, and therapeutics. References 2007. Hematological effects of four ethylene glycol monoalkyl ethers in short-term repeated exposure in rats. Archives of Toxicology, 82(2). DOI: 10.1007/s00204-007-0236-z 2004. Exposure to glycol ethers in a population of French men evaluated by measurement of urinary alkoxycarboxylic acids. International Archives of Occupational and Environmental Health, 77(5). DOI: 10.1007/s00420-004-0523-4 1991. Gas chromatographic determination of butoxyacetic acid in human blood after exposure to 2-butoxyethanol. Archives of Toxicology, 65(5). DOI: 10.1007/bf02284269 |
| Market Analysis Reports |