Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Lontr Science & Technology Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lontr.com | |||
 | +86 (25) 8434-1022 | |||
 | +86 (25) 8434-1022 | |||
 | zjb298680@163.com zjb29868@hotmail.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Xi'an Tuochao Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xatuochao.cn | |||
 | +86 18092957403 | |||
 | chen.zeshan@xatuochao.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: +8618092957403 | |||
 | WhatsApp:+8618092957403 | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| ECA International Corporation | USA | |||
|---|---|---|---|---|
 | www.ecacorporation.com | |||
 | +1 (847) 358-8178 | |||
 | +1 (847) 358-8179 | |||
 | order@ecacorporation.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Frinton Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.frinton.com | |||
 | +1 (856) 722-7037 | |||
 | +1 (856) 439-1977 | |||
 | sales@frinton.com frinton@frinton.com | |||
| Chemical manufacturer since 1963 | ||||
| Classification | Organic raw materials >> Hydrocarbon compounds and their derivatives >> Hydrocarbon nitrite |
|---|---|
| Name | 2-Methyl-3-nitrophenol |
| Synonyms | 2-Hydroxy-6-nitrotoluene; 3-Nitro-o-cresol |
| Molecular Structure | 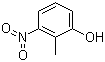 |
| Molecular Formula | C7H7NO3 |
| Molecular Weight | 153.13 |
| CAS Registry Number | 5460-31-1 |
| EC Number | 226-739-0 |
| SMILES | CC1=C(C=CC=C1O)[N+](=O)[O-] |
| Density | 1.3±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 147-152 °C |
| Index of Refraction | 1.597, Calc.* |
| Boiling Point | 269.5±28.0 °C (760 mmHg), Calc.* |
| Flash Point | 121.8±12.5 °C, Calc.* |
| Hazard Symbols |   GHS06;GHS07 Danger Details GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H311-H312-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P262-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P316-P317-P319-P321-P330-P332+P317-P337+P317-P361+P364-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2446 | ||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||
|
2-Methyl-3-nitrophenol, also known as 3-nitro-o-cresol, is an organic compound characterized by a nitro group and a methyl group attached to a phenolic ring. With the chemical formula C7H7N O3, this compound is notable for its unique chemical properties and its varied applications in different industries. The discovery of 2-methyl-3-nitrophenol dates back to the early 20th century, during a period marked by extensive research into nitrophenolic compounds, which were of great interest due to their potential applications in dyes, pharmaceuticals, and agrochemicals. The synthesis of 2-methyl-3-nitrophenol typically involves the nitration of o-cresol, a process that introduces a nitro group into the aromatic ring. This reaction can be conducted using a mixture of nitric acid and sulfuric acid under controlled conditions to ensure the proper substitution pattern. The positioning of the nitro group at the 3-position relative to the methyl group is crucial, as it influences the compound's reactivity and its behavior in subsequent reactions. The resulting product is a yellowish crystalline solid with a distinct odor, which is soluble in organic solvents. One of the primary applications of 2-methyl-3-nitrophenol is as an intermediate in the production of dyes and pigments. Its ability to undergo various chemical transformations makes it a valuable building block in the synthesis of more complex organic compounds. The nitro group can be further reduced or transformed to create a range of dye products, which find use in textiles, inks, and plastics. The compound's structure allows for the development of azo dyes, which are widely used due to their vibrant colors and stability. In addition to its role in dye production, 2-methyl-3-nitrophenol is utilized in the synthesis of pharmaceuticals. Its chemical reactivity enables it to participate in various coupling reactions, leading to the formation of biologically active compounds. Researchers have investigated the potential of derivatives of 2-methyl-3-nitrophenol in medicinal chemistry, particularly for their antimicrobial and antifungal properties. These investigations highlight the compound's significance in the development of new therapeutic agents. Furthermore, 2-methyl-3-nitrophenol has been studied for its role as a pesticide and herbicide. Its effectiveness against certain pests has led to its incorporation into formulations designed to protect crops from insect damage. The compound's toxicity to specific organisms underscores the importance of understanding its environmental impact and the need for careful handling during agricultural applications. Despite its usefulness, the application of 2-methyl-3-nitrophenol is not without challenges. The compound, like many nitrophenols, has been associated with environmental concerns due to its potential toxicity and persistence in ecosystems. Consequently, regulatory frameworks have been established to manage its use and mitigate potential risks to human health and the environment. Ongoing research aims to develop safer alternatives and improve the sustainability of practices involving this compound. In conclusion, 2-methyl-3-nitrophenol is an important chemical compound with diverse applications in dye production, pharmaceuticals, and agriculture. Its unique chemical properties and reactivity make it a valuable intermediate in various synthetic processes. As industries continue to evolve, the exploration of 2-methyl-3-nitrophenol's potential will likely expand, contributing to advancements in materials science, medicinal chemistry, and agrochemical formulations. References 2007. Identification of highly polar nitroaromatic compounds in leachate and ground water samples from a TNT-contaminated waste site by LC-MS, LC-NMR, and off-line NMR and MS investigations. Analytical and Bioanalytical Chemistry, 389(6). DOI: 10.1007/s00216-007-1573-8 2005. Spatial and Geographical Variations of Urban, Suburban and Rural Atmospheric Concentrations of Phenols and Nitrophenols. Environmental Science and Pollution Research, 12(6). DOI: 10.1065/espr2005.06.264 2007. Solid-phase microextraction and gas chromatography-mass spectrometry for analysis of phenols and nitrophenols in rainwater, as their t-butyldimethylsilyl derivatives. Analytical and Bioanalytical Chemistry, 387(4). DOI: 10.1007/s00216-006-1115-9 |
| Market Analysis Reports |