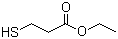
Ethyl 3-mercaptopropionate is an organic compound belonging to the class of thiol esters. It is composed of an ethyl ester group (-COOCH2CH3) attached to the sulfur-containing mercaptopropionate moiety (C3H5O2S). The structure consists of a three-carbon chain, where the central carbon is bonded to a thiol group (-SH) at the 3-position, and the terminal carboxyl group is esterified with an ethyl group.
This compound is significant due to the presence of both the mercapto group (-SH) and the ester functional group, which imparts unique reactivity and properties to the molecule. The mercapto group is highly nucleophilic, making it reactive in a wide range of chemical reactions. This reactivity is often exploited in the synthesis of various other chemical compounds, particularly in processes involving thiol-based nucleophilic substitution or thiol-disulfide exchange reactions.
Ethyl 3-mercaptopropionate has several applications in different chemical and industrial sectors. It is commonly used as a building block in the synthesis of various pharmaceuticals, agrochemicals, and specialty chemicals. Its thiol group makes it a useful precursor in the production of thiol-functionalized materials, such as polymers and other materials that require sulfur-containing functional groups.
In addition to its use in organic synthesis, ethyl 3-mercaptopropionate has been studied for its potential biological activities. Thiol esters, such as this compound, are often investigated for their antioxidant, anti-inflammatory, and antimicrobial properties. The mercapto group plays a key role in mediating such activities by interacting with free radicals or by forming coordination complexes with metal ions, which may enhance the compound's biological effects.
Furthermore, ethyl 3-mercaptopropionate can be employed in the preparation of thiol-based reagents for use in the preparation of peptide-based compounds or in other synthetic chemistry reactions that require thiol groups for cross-linking or modification.
In the field of materials science, the sulfur-containing structure of ethyl 3-mercaptopropionate also makes it an important component for the design of functional materials. It can be used in the synthesis of materials with self-assembling or adhesive properties, or in the modification of surfaces to improve their chemical resistance or stability.
Overall, ethyl 3-mercaptopropionate is a versatile compound with numerous applications in organic synthesis, biological studies, and materials science, driven by the reactivity and versatility of its thiol and ester groups.
|

























 GHS07 Warning Details
GHS07 Warning Details