Online Database of Chemicals from Around the World
| Nanjing Carbonde Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.carbonde.com | |||
 | +86 (25) 5857-3173 | |||
 | +86 (25) 8347-5170 | |||
 | sales@carbonde.com wuq_11@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Shanghai Benrochem Chemcal Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.benrochem.com.cn | |||
 | +86 13917534498 | |||
 | +86 (21) 5217-8644 | |||
 | johnszcn@hotmail.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2010 | ||||
| Labseeker Inc | USA | |||
|---|---|---|---|---|
 | www.labseeker.com | |||
 | +1 (858) 750-1632 | |||
 | +1 (858) 412-1220 | |||
 | marketing@labseeker.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| ProChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.prochemonline.com | |||
 | +1 (815) 398-1788 | |||
 | +1 (815) 398-1810 | |||
 | prochem3@aol.com | |||
| Chemical manufacturer since 1986 | ||||
| Classification | Organic raw materials >> Organometallic compound >> Organic copper |
|---|---|
| Name | Copper(I) bromide-dimethyl sulfide |
| Molecular Structure | 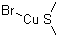 |
| Molecular Formula | C2H6BrCuS |
| Molecular Weight | 205.58 |
| CAS Registry Number | 54678-23-8 |
| EC Number | 259-292-5 |
| SMILES | CSC.[Cu]Br |
| Melting point | 132 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315 Details | ||||||||||||||||||||
| Safety Statements | P280-P305+P351+P338 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Copper(I) bromide-dimethyl sulfide, often abbreviated as CuBr·DMS, is a significant coordination complex with various applications in organic synthesis and catalysis. The compound features copper(I) bromide coordinated with dimethyl sulfide (DMS), a combination that has proven useful in a range of chemical processes. The discovery and applications of Copper(I) bromide-dimethyl sulfide illustrate its role in advancing both synthetic chemistry and industrial applications. The synthesis of Copper(I) bromide-dimethyl sulfide involves the coordination of dimethyl sulfide to Copper(I) bromide. The reaction typically occurs in an inert atmosphere to prevent oxidation of the copper(I) center. This complex is prepared by mixing Copper(I) bromide with dimethyl sulfide under controlled conditions, leading to the formation of a stable coordination compound. The dimethyl sulfide acts as a ligand that stabilizes the copper(I) center, enhancing its utility in various catalytic reactions. Structurally, Copper(I) bromide-dimethyl sulfide consists of a Copper(I) ion coordinated to a bromide ion and dimethyl sulfide ligand. The copper(I) ion, which is in the +1 oxidation state, is a key component in many catalytic processes due to its ability to facilitate electron transfer and participate in oxidation-reduction reactions. The dimethyl sulfide ligand stabilizes the copper(I) center and influences its reactivity. One of the primary applications of Copper(I) bromide-dimethyl sulfide is in organic synthesis, particularly in coupling reactions. The compound is used as a catalyst in various cross-coupling reactions, such as the Ullmann coupling and the Sonogashira coupling. In these reactions, Copper(I) bromide-dimethyl sulfide facilitates the formation of carbon-carbon bonds by mediating the transfer of electrons between reactants. This application is crucial for the synthesis of complex organic molecules, including pharmaceuticals and fine chemicals. In addition to coupling reactions, Copper(I) bromide-dimethyl sulfide is employed in other catalytic processes. For example, it is used in the oxidation of alcohols to aldehydes or ketones. The compound's ability to participate in electron transfer processes makes it a valuable catalyst for these transformations, which are important in the synthesis of various organic compounds. Copper(I) bromide-dimethyl sulfide also finds applications in the field of material science. The compound is used in the preparation of organic semiconductors and light-emitting materials. Its role as a catalyst in the synthesis of these materials contributes to the development of advanced electronic devices and displays. The stability and reactivity of Copper(I) bromide-dimethyl sulfide make it a useful tool in both laboratory and industrial settings. The compound should be handled with care to avoid exposure to air and moisture, which can lead to the oxidation of the copper(I) center and degradation of the complex. Proper storage in a dry, inert atmosphere is essential to maintain its effectiveness. Future research on Copper(I) bromide-dimethyl sulfide may focus on improving its catalytic efficiency and exploring new applications. Advances in understanding its coordination chemistry and reactivity could lead to the development of more effective catalysts and new synthetic methodologies. References 1991. Synthesis of macrolide antibiotics. 22. Synthesis of the C8-C13 fragment of oleandonolide. Bulletin of the Academy of Sciences of the USSR, Division of chemical science, 40(9). DOI: 10.1007/bf00960425 1985. Selenoesters in organic synthesis. 2. New synthesis of saturated and α,β-unsaturated ketones and the synthesis of the Peach moth pheromone (Carposia niponensis). Bulletin of the Academy of Sciences of the USSR, Division of chemical science, 34(7). DOI: 10.1007/bf00950160 1983. Synthesis of (Z,Z)-3, 13-octadecadien-1-yl acetate Component of the sex pheromone of Synanthedon tenuis. Journal of Chemical Ecology, 9(2). DOI: 10.1007/bf00988040 |
| Market Analysis Reports |