Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Xuchang Chenhe Bio-pharmaceutical Tech. Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.the-chem.com | |||
 | +86 (374) 579-3128 +86 15836530076 | |||
 | +86 (374) 579-3128 | |||
 | wxh5168@126.com wxh5168@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Ningbo Syntame Biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.syntame.com | |||
 | +86 (574) 2880-2872 | |||
 | sales@syntame.com | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| ECA International Corporation | USA | |||
|---|---|---|---|---|
 | www.ecacorporation.com | |||
 | +1 (847) 358-8178 | |||
 | +1 (847) 358-8179 | |||
 | order@ecacorporation.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Ketone compound |
|---|---|
| Name | (S)-(-)-4-tert-Butyl-2-oxazolidinone |
| Synonyms | (4S)-4-tert-butyl-1,3-oxazolidin-2-one |
| Molecular Structure | 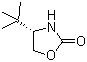 |
| Molecular Formula | C7H13NO2 |
| Molecular Weight | 143.18 |
| CAS Registry Number | 54705-42-9 |
| EC Number | 828-370-6 |
| SMILES | CC(C)(C)[C@H]1COC(=O)N1 |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
(S)-(-)-4-tert-Butyl-2-oxazolidinone is an enantiomerically pure compound widely used in organic synthesis, particularly in asymmetric synthesis and chiral resolution. Its oxazolidinone core is a versatile scaffold, and the presence of the tert-butyl group at the 4-position enhances steric and electronic effects, making it a highly effective auxiliary in stereoselective transformations. This compound has played a significant role in advancing methodologies for the synthesis of enantiomerically enriched molecules. The discovery of (S)-(-)-4-tert-butyl-2-oxazolidinone can be attributed to the search for efficient chiral auxiliaries in asymmetric synthesis. Researchers focused on oxazolidinones due to their rigid five-membered ring structure, which enables precise control of stereochemistry in subsequent reactions. The introduction of a bulky tert-butyl group at the 4-position was found to improve selectivity by providing steric hindrance and minimizing undesired pathways. The (S)-configuration was specifically designed to cater to reactions requiring enantiomeric control. In synthetic organic chemistry, (S)-(-)-4-tert-butyl-2-oxazolidinone is extensively used as a chiral auxiliary in aldol reactions, Michael additions, and Diels-Alder reactions. Its ability to influence the stereochemical outcome of these reactions is attributed to its steric bulk and the conformational rigidity of its structure. For example, in aldol reactions, the oxazolidinone moiety directs the approach of electrophiles and nucleophiles to achieve high diastereoselectivity, leading to the preferential formation of a single enantiomer of the product. This compound has also found applications in the preparation of natural products and pharmaceutical intermediates. It has been utilized in the synthesis of complex molecules, such as alkaloids and amino acids, where controlling stereochemistry is critical for biological activity. Its use as a chiral auxiliary has simplified synthetic routes, reducing the number of steps and improving overall yields. In addition to its applications in asymmetric synthesis, (S)-(-)-4-tert-butyl-2-oxazolidinone has been employed in the resolution of racemic mixtures. By forming diastereomeric derivatives, it facilitates the separation of enantiomers, enabling the isolation of optically pure compounds. This method is particularly valuable in the pharmaceutical industry, where the production of enantiomerically pure drugs is essential for regulatory approval and therapeutic efficacy. The compound’s stability, ease of recovery, and reusability further enhance its appeal as a chiral auxiliary. After the desired reaction is complete, the oxazolidinone can often be regenerated, reducing waste and making it an environmentally friendly choice in synthetic chemistry. (S)-(-)-4-tert-Butyl-2-oxazolidinone continues to be an indispensable tool in modern organic synthesis, enabling chemists to achieve precision and efficiency in the preparation of chiral molecules. Its contributions to asymmetric synthesis and chiral resolution underscore its importance in both academic research and industrial applications. References 2023. Synthesis and Biochemical Evaluation of Monocarboxylic GRB2 SH2 Domain Inhibitors. Methods in molecular biology (Clifton, N.J.). DOI: 10.1007/978-1-0716-3393-9_15 2022. Recent Advances in Enantioselective Organocatalytic Reactions Enabled by N-Heterocyclic Carbenes (NHCs) Containing Triazolium Motifs. Synthesis, 54(15). DOI: 10.1055/a-1856-5688 2019. Synthetic Approaches to Chiral Non-C 2-symmetric N-Heterocyclic Carbene Precursors. Synthesis, 51(06). DOI: 10.1055/s-0037-1611733 |
| Market Analysis Reports |