Online Database of Chemicals from Around the World
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Classification | Biochemical >> Inhibitor |
|---|---|
| Name | N-(2-Chloroethyl)dibenzylamine hydrochloride |
| Synonyms | N,N-dibenzyl-2-chloroethanamine;hydrochloride |
| Molecular Structure | 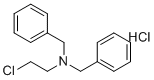 |
| Molecular Formula | C16H19Cl2N |
| Molecular Weight | 296.23 |
| CAS Registry Number | 55-43-6 |
| EC Number | 200-234-5 |
| SMILES | C1=CC=C(C=C1)CN(CCCl)CC2=CC=CC=C2.Cl |
| Melting Point | 190-193 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
N-(2-Chloroethyl)dibenzylamine hydrochloride is an important compound in organic chemistry and medicinal research. Its structure comprises a dibenzylamine core with a chloroethyl group, and it is often used in various chemical and pharmaceutical applications due to its unique properties. The compound was first synthesized as part of efforts to develop new chemical reagents and intermediates. The presence of the chloroethyl group allows it to participate in a range of chemical reactions, making it a versatile building block in organic synthesis. This functionality is particularly useful in the synthesis of complex organic molecules and pharmaceuticals. In pharmaceutical chemistry, N-(2-Chloroethyl)dibenzylamine hydrochloride has found applications in the development of new drugs. The chloroethyl group is known for its reactivity and can be used to create derivatives that have potential therapeutic effects. The compound has been investigated for its role in designing drugs that target specific biological pathways or exhibit particular pharmacological activities. One significant application of N-(2-Chloroethyl)dibenzylamine hydrochloride is in the synthesis of antitumor agents. The chloroethyl group is a key component in the structure of several chemotherapeutic agents, and the compound serves as an intermediate in the preparation of such drugs. By modifying the dibenzylamine structure, researchers can develop compounds with enhanced efficacy and reduced side effects for cancer treatment. Additionally, this compound is employed as a reagent in organic synthesis for various transformations. Its ability to participate in nucleophilic substitution reactions makes it valuable in creating new chemical entities. The versatility of N-(2-Chloroethyl)dibenzylamine hydrochloride extends to its use in the synthesis of agrochemicals, dyes, and other specialty chemicals. In summary, N-(2-Chloroethyl)dibenzylamine hydrochloride is a significant compound in both pharmaceutical and organic chemistry. Its unique structure allows it to be used as an intermediate in drug synthesis and a reagent in chemical transformations. Its applications range from drug development to the production of specialty chemicals, highlighting its importance in chemical research and industry. References 1976. Synthesis and adrenoreceptor blocking action of aziridinium ions derived from phenoxybenzamine and dibenamine. Journal of Medicinal Chemistry. DOI: 1536 1983. Muscarinic receptors of rabbit irides. Naunyn-Schmiedeberg's Archives of Pharmacology. DOI: 6866127 1999. Experimental and theoretical comparisons between the classical Schild analysis and a new alternative method to evaluate the pA2 of competitive antagonists. Naunyn-Schmiedeberg's Archives of Pharmacology. DOI: 10598787 |
| Market Analysis Reports |