Online Database of Chemicals from Around the World
| ChemPacific Corp | USA | |||
|---|---|---|---|---|
 | www.chempacific.com | |||
 | +1 (410) 633-5771 | |||
 | +1 (410) 633-5808 | |||
 | sales@chempacific.com | |||
| Chemical manufacturer since 1995 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Nanjing Bic Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bicbiotech.com | |||
 | +86 18851183712 | |||
 | sales@bicbiotech.com | |||
 | QQ Chat | |||
 | WeChat: +8618851183712 | |||
 | WhatsApp:+8618851183712 | |||
| Chemical manufacturer since 2023 | ||||
| chemBlink Standard supplier since 2026 | ||||
| AK Scientific, Inc | USA | |||
|---|---|---|---|---|
 | www.aksci.com | |||
 | +1 (510) 429-8835 | |||
 | +1 (510) 429-8836 | |||
 | sales@aksci.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Piperidines |
|---|---|
| Name | 4-(Morpholin-4-yl)-piperidine dihydrochloride |
| Synonyms | 4-piperidin-4-ylmorpholine;dihydrochloride |
| Molecular Structure | 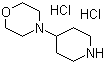 |
| Molecular Formula | C9H18N2O.2(HCl) |
| Molecular Weight | 243.17 |
| CAS Registry Number | 550370-31-5 |
| SMILES | C1CNCCC1N2CCOCC2.Cl.Cl |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319-H335 Details |
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details |
| SDS | Available |
|
4-(Morpholin-4-yl)-piperidine dihydrochloride is a salt form of a bifunctional heterocyclic compound in which a piperidine ring is substituted at the 4-position with a morpholine moiety. The dihydrochloride form results from protonation of the two nitrogen atoms—one in the piperidine and one in the morpholine ring—providing enhanced water solubility and improved chemical stability, which is particularly useful for handling, storage, and formulation in research and pharmaceutical applications. This structural arrangement combines the basicity and conformational flexibility of the piperidine ring with the polar, hydrogen-bonding characteristics of morpholine, creating a scaffold widely used in medicinal chemistry. Structurally, the molecule features a six-membered saturated piperidine ring connected at its 4-position to a morpholine ring, which contains an oxygen atom within the ring system. Both nitrogen atoms are protonated in the dihydrochloride salt, forming stable ionic interactions with chloride ions. The spatial arrangement of the two rings offers conformational flexibility while maintaining a defined geometry, allowing favorable interactions with biological targets such as enzymes, receptors, and transporters. The polar and basic nature of the molecule also facilitates formulation in aqueous systems. The preparation of 4-(Morpholin-4-yl)-piperidine dihydrochloride typically involves first synthesizing 4-(morpholin-4-yl)piperidine through nucleophilic substitution of a 4-halopiperidine with morpholine under controlled conditions. Subsequent treatment with hydrochloric acid converts the free base into the dihydrochloride salt. This conversion improves water solubility, reduces volatility, and stabilizes the compound, making it suitable for use in biological assays or as an intermediate for further synthetic transformations. In medicinal chemistry, this compound serves as a versatile building block for drug design. The protonated piperidine nitrogen can engage in ionic interactions with acidic residues on protein surfaces, while the morpholine moiety can form hydrogen bonds and improve aqueous solubility. These properties allow chemists to design derivatives targeting central nervous system receptors, transporters, or enzymes. The dihydrochloride salt ensures consistent behavior in solution and facilitates accurate dosing in pharmacological studies. Beyond drug design, 4-(Morpholin-4-yl)-piperidine dihydrochloride is widely used as a synthetic intermediate. The protonated nitrogen atoms can be selectively deprotonated and functionalized, enabling modifications such as acylation, alkylation, or cross-coupling reactions. The bifunctional heterocyclic scaffold allows chemists to introduce diverse substituents at either ring, producing libraries of derivatives for structure–activity relationship studies or as precursors for more complex heterocyclic systems. Overall, 4-(Morpholin-4-yl)-piperidine dihydrochloride exemplifies a bifunctional heterocyclic salt with enhanced solubility and stability. Its combination of a basic piperidine ring, a polar morpholine ring, and the dihydrochloride form makes it valuable as a building block in medicinal chemistry, organic synthesis, and the development of biologically active compounds. References 2016. Substituted pyridines as inhibitors of DNMT1. EP Patent. URL: EP-3468953-A1 2016. Substituted pyridines as inhibitors of DNMT1. CA Patent. URL: CA-3026226-A1 2016. Substituted pyridines as inhibitors of DNMT1 (granted). US Patent. URL: US-10975056-B2 2007. New preparation method. CN Patent. URL: CN-101821258-A |
| Market Analysis Reports |