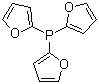
Tri(2-furyl)phosphine is a notable phosphine ligand widely recognized for its applications in organometallic chemistry and catalysis. The compound, consisting of a phosphorus atom bonded to three 2-furyl groups, plays a significant role in various chemical reactions due to its unique electronic and steric properties. This article discusses the discovery and applications of Tri(2-furyl)phosphine, highlighting its importance in modern chemistry.
Tri(2-furyl)phosphine was first synthesized in the mid-20th century as part of research into phosphine ligands for catalytic processes. The synthesis typically involves the reaction of phosphorus trichloride with 2-furyl lithium or other 2-furyl derivatives, resulting in the formation of the desired tri(2-furyl)phosphine. The resulting ligand features a phosphorus atom centrally bonded to three furyl rings, which are aromatic systems containing both carbon and oxygen atoms. This structure imparts distinctive properties to the ligand, including electron-donating effects and coordination flexibility.
One of the primary applications of Tri(2-furyl)phosphine is in catalytic processes. The ligand is used in a variety of catalytic reactions, particularly in transition metal complexes. Tri(2-furyl)phosphine forms stable complexes with metals such as palladium, platinum, and rhodium. These metal-ligand complexes are employed in numerous catalytic reactions, including hydrogenation, hydroformylation, and cross-coupling reactions. The electron-donating properties of the furyl groups can influence the electronic environment of the metal center, enhancing catalytic activity and selectivity.
In addition to its role in traditional catalytic reactions, Tri(2-furyl)phosphine is also used in the synthesis of complex organic molecules. For example, it has been employed in the preparation of pharmaceutical intermediates and fine chemicals. The ligand's ability to form stable complexes with transition metals facilitates the formation of carbon-carbon and carbon-heteroatom bonds, which are crucial for the synthesis of diverse organic compounds.
Tri(2-furyl)phosphine is also significant in the field of organometallic chemistry due to its ability to stabilize various oxidation states of metal centers. The ligand's coordination properties allow it to stabilize metals in lower oxidation states, which can be advantageous for certain catalytic processes. This stabilization capability can lead to the development of new catalytic systems with improved efficiency and selectivity.
The unique electronic and steric properties of Tri(2-furyl)phosphine also make it valuable in the study of reaction mechanisms and coordination chemistry. Researchers use this ligand to investigate the behavior of metal complexes in different chemical environments, providing insights into reaction pathways and mechanisms.
Handling Tri(2-furyl)phosphine requires standard precautions to avoid exposure to air and moisture, which can lead to degradation of the ligand. Proper storage in a dry, inert atmosphere is essential to maintain its stability and reactivity.
Future research on Tri(2-furyl)phosphine may focus on exploring new applications and improving its performance in catalytic processes. Advances in understanding its coordination chemistry and reactivity could lead to the development of more efficient and selective catalysts for various chemical transformations.
References
2024. Cationic palladium cyclopentadienyl complexes with tri(2-furyl)phosphine for norbornene polymerization. Kinetics and Catalysis, 65(1).
DOI: 10.1134/s0023158424010087
2021. Structure and catalytic properties of palladium complex with tri(2-furyl)phosphine. Journal of Structural Chemistry, 62(8).
DOI: 10.1134/s0022476621080072
|























































 GHS07 Warning Details
GHS07 Warning Details