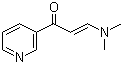
1-(3-Pyridyl)-3-(dimethylamino)-2-propen-1-one is a functionalized α,β-unsaturated ketone derivative in which a 3-pyridyl group is conjugated with a dimethylamino-substituted propenone system. Compounds of this type are of interest in organic and medicinal chemistry due to their electrophilic α,β-unsaturated carbonyl functionality, which allows Michael-type additions and nucleophilic conjugate reactions, as well as their heteroaryl substitution, which provides hydrogen bonding, π–π interactions, and electron-withdrawing effects. The dimethylamino group contributes both electron-donating resonance effects and steric characteristics, influencing reactivity and biological potential.
Structurally, the molecule consists of a three-carbon α,β-unsaturated ketone backbone in which the carbonyl carbon is attached to a 3-pyridyl ring, and the β-carbon bears a dimethylamino group. The pyridine ring contains a nitrogen atom at the meta position relative to the carbonyl, which increases polarity and allows for potential interactions with metal ions or enzyme active sites. The conjugation between the ketone and the pyridine ring enhances the electrophilicity of the β-carbon, making it susceptible to nucleophilic attack in synthetic applications.
The synthesis of 1-(3-pyridyl)-3-(dimethylamino)-2-propen-1-one is typically achieved via condensation reactions between 3-acetylpyridine and dimethylamino-substituted aldehydes or equivalents under basic or acidic catalysis. The α,β-unsaturated system is formed through an aldol-type condensation or Knoevenagel reaction, allowing the controlled installation of the dimethylamino group at the β-position. These methods provide selective formation of the (E)- or (Z)-isomer depending on reaction conditions, ensuring the desired stereochemistry for further reactions or applications.
In medicinal chemistry, α,β-unsaturated ketones with heteroaryl substitution are explored as electrophilic pharmacophores capable of interacting with nucleophilic residues in enzymes or receptors, forming covalent or non-covalent complexes. The 3-pyridyl ring can engage in hydrogen bonding and π-stacking interactions, enhancing binding affinity and selectivity. The dimethylamino substituent modulates the electronic density of the conjugated system and may improve solubility and lipophilicity, allowing optimization of pharmacokinetic and pharmacodynamic properties.
Beyond pharmaceutical applications, this compound is valuable as a synthetic intermediate in organic chemistry. The α,β-unsaturated carbonyl system can participate in Michael additions, cycloadditions, or nucleophilic substitutions, enabling the construction of complex heterocyclic frameworks, substituted pyridines, or conjugated enone derivatives. The dimethylamino group can also be modified through N-alkylation or quaternization, expanding the structural diversity accessible from this scaffold.
Overall, 1-(3-Pyridyl)-3-(dimethylamino)-2-propen-1-one represents a bifunctional electrophilic and heteroaryl-substituted enone. Its combination of a 3-pyridyl ring, conjugated α,β-unsaturated carbonyl, and a dimethylamino group provides chemical versatility for nucleophilic additions, heterocyclic synthesis, and medicinal chemistry applications, making it a valuable intermediate and building block for the development of bioactive compounds.
References
2021. Acyloxylation of Enaminones. Science of Synthesis.
URL: SD-132-00312
|



















































 GHS07;GHS08 Warning Details
GHS07;GHS08 Warning Details