Online Database of Chemicals from Around the World
| Sinocompound Catalysts Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinocompound.com | |||
 | +86 (512) 6721-6630 | |||
 | +86 (512) 5631-6689 | |||
 | sales@sinocompound.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BeiJing VsciChem Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.vscichem.com | |||
 | +86 (10) 5248-3976 | |||
 | +86 (10) 6027-0130 | |||
 | vscichem@163.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Rhodia (shanghai) International Trading Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rhodia.com | |||
 | +86 (21) 2408-9317 +86 13601753458 | |||
 | +86 (21) 5483-0035 | |||
 | hank.xue@ap.rhodia.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Ereztech LLC | USA | |||
|---|---|---|---|---|
 | www.ereztech.com | |||
 | +1 (888) 658-1221 | |||
 | sales@ereztech.com | |||
| Chemical distributor since 2010 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Organic phosphine compound |
|---|---|
| Name | Triethylphosphine |
| Molecular Structure | 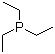 |
| Molecular Formula | C6H15P |
| Molecular Weight | 118.16 |
| CAS Registry Number | 554-70-1 |
| EC Number | 209-068-8 |
| SMILES | CCP(CC)CC |
| Density | 0.802 |
|---|---|
| Melting Point | -88 °C |
| Boiling point | 127-128 °C |
| Refractive index | 1.456 |
| Flash point | -17.2222 °C |
| Hazard Symbols |    GHS02;GHS05;GHS07 Danger Details GHS02;GHS05;GHS07 Danger Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H224-H225-H250-H302-H314-H318 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P210-P222-P231-P233-P240-P241-P242-P243-P260-P264-P264+P265-P270-P280-P301+P317-P301+P330+P331-P302+P335+P334-P302+P361+P354-P303+P361+P353-P304+P340-P305+P354+P338-P316-P317-P321-P330-P363-P370+P378-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| Transport Information | UN 2845 | ||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
Triethylphosphine is a versatile organophosphorus compound recognized for its significant role in various chemical applications. This compound, with the chemical formula (C2H5)3P, features a phosphorus atom bonded to three ethyl groups. Its discovery and subsequent applications underscore its importance in the fields of catalysis, coordination chemistry, and organic synthesis. This article provides an overview of the discovery, applications, and utility of triethylphosphine in modern chemical research and industry. Triethylphosphine was first synthesized in the early 20th century as chemists explored new organophosphorus compounds. The compound is typically prepared by the reaction of phosphorus trichloride with ethanol or ethyl alcohol in the presence of a base, such as sodium hydroxide. This synthesis yields triethylphosphine as the primary product, which can then be purified and used for various applications. The straightforward synthesis and the stable nature of triethylphosphine have made it a valuable reagent in both academic and industrial settings. One of the primary applications of triethylphosphine is in the field of catalysis. The compound acts as a ligand in the formation of metal complexes, particularly with transition metals such as palladium, platinum, and nickel. These metal-ligand complexes are crucial for numerous catalytic processes, including cross-coupling reactions, hydrogenation, and carbonylation. Triethylphosphine is favored for its ability to stabilize metal centers and influence the electronic properties of the metal, leading to improved catalytic efficiency and selectivity. In coordination chemistry, triethylphosphine is used to synthesize a variety of metal complexes with different coordination environments. Its three ethyl groups provide a significant steric bulk, which affects the geometry and reactivity of the resulting metal complexes. This steric influence is valuable in designing complexes with specific properties, such as enhanced reactivity or selectivity for particular reactions. Triethylphosphine also finds application in organic synthesis, where it serves as a nucleophile and a reducing agent. Its ability to participate in nucleophilic substitution reactions makes it useful for modifying organic molecules and constructing complex structures. Additionally, triethylphosphine can act as a reducing agent in various chemical transformations, including the reduction of organic halides and other functional groups. The compound's properties include its volatility and relatively low toxicity, making it convenient for use in laboratory settings. Triethylphosphine is generally handled with standard safety precautions, including working in a well-ventilated area and using appropriate personal protective equipment. Proper storage conditions are also essential to maintain the stability of the compound, typically in a cool, dry place. Future research on triethylphosphine may explore its potential in developing new catalytic systems and materials. Advances in understanding its interaction with different metal centers and its behavior in various chemical environments could lead to innovative applications and enhanced performance in existing catalytic and synthetic processes. References 2024. On Phosphine-containing Gold(I) Complexes in Solution and Their Biological Application. Russian Journal of Inorganic Chemistry. DOI: 10.1134/s0036023624600965 2023. Synthesis, Structure, and Catalytic Properties of Palladium(II) bis(diphenylcyclohexylphosphine) (ζ5-cyclopentadienyl) Tetrafluoroborate in Butadiene Telomerization with Methanol. Journal of Structural Chemistry. DOI: 10.1134/s0022476623110203 2023. Reaction of 2-R-naphtho[2,3-d][1,3,2]dioxaphosphinin-4-ones with arylidene derivatives of malonic acid esters: synthesis, molecular and crystal structures of 5-oxo-2-R-naphtho[2,3-f][1,2]oxaphosphepine 2-oxides. Russian Chemical Bulletin. DOI: 10.1007/s11172-023-3865-2 |
| Market Analysis Reports |