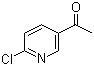
2-Chloro-5-acetylpyridine is a halogenated pyridine derivative featuring a chlorine atom at the 2-position and an acetyl group at the 5-position of the pyridine ring. This compound belongs to a class of heteroaromatic ketones that are frequently used as building blocks and intermediates in the synthesis of more complex organic molecules, particularly within pharmaceutical and agrochemical contexts.
The structural features of 2-chloro-5-acetylpyridine—namely the electron-deficient pyridine ring, the electron-withdrawing chlorine atom, and the reactive ketone moiety—confer valuable chemical reactivity. The acetyl group at the 5-position introduces a carbonyl function capable of undergoing typical ketone transformations such as nucleophilic addition, condensation, and enolate formation. The 2-chloro substituent enhances the ring's reactivity towards nucleophilic aromatic substitution (SNAr), especially under basic or catalytic conditions, making it a suitable precursor for the introduction of various nucleophiles.
The discovery and utilization of this compound stem from broader research into substituted pyridines as pharmacophores. Pyridine derivatives, including halogenated and acylated analogs, have been recognized for their biological activity and their capacity to mimic natural substrates or ligands in biological systems. In particular, the 2-chloro substituent on the pyridine ring facilitates synthetic derivatization through SNAr reactions, while the 5-acetyl group can serve as a handle for further functionalization, making 2-chloro-5-acetylpyridine a highly adaptable intermediate.
In medicinal chemistry, 2-chloro-5-acetylpyridine has been employed in the synthesis of small molecules designed to target various enzymes and receptors. The pyridine ring often serves as a key structural element that can engage in π–π stacking, hydrogen bonding, and other interactions within biological binding sites. Modifications at the acetyl group allow medicinal chemists to introduce additional heterocycles or to create fused ring systems that enhance pharmacokinetic properties or specificity.
Beyond pharmaceuticals, this compound has seen application in agrochemical development. Substituted pyridines are common in herbicides and insecticides due to their capacity to disrupt biological processes in target organisms while maintaining selectivity and stability. The reactivity of the acetyl group enables the synthesis of analogs with modified lipophilicity, metabolic resistance, or improved bioavailability for environmental use.
Synthetically, 2-chloro-5-acetylpyridine can be prepared via selective halogenation and acylation strategies. One typical route involves the Friedel–Crafts acylation of 2-chloropyridine derivatives, although regioselective control is necessary to ensure functionalization at the 5-position. Alternatively, stepwise functionalization starting from 5-bromopyridine can lead to the target compound through sequential substitution and ketone formation steps.
The ketone moiety of 2-chloro-5-acetylpyridine also lends itself to applications in coordination chemistry. The carbonyl group can act as a ligand or be converted into oximes, hydrazones, or enaminones, which in turn can serve as chelating agents in transition metal complexes. These complexes may exhibit catalytic activity or serve in material science applications.
Overall, 2-chloro-5-acetylpyridine plays an important role in modern synthetic chemistry due to its bifunctional reactivity and its compatibility with numerous derivatization pathways. It continues to be a useful tool in the development of novel compounds for pharmaceutical, agricultural, and materials science applications.
References
2023. A pharmacophore-based approach to demonstrating the scope of alcohol dehydrogenases. Bioorganic & Medicinal Chemistry, 82.
DOI: 10.1016/j.bmc.2023.117255
2014. With Acid Chlorides. Science of Synthesis, 1.
URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-020-00412
|
























































 GHS06;GHS07 Danger Details
GHS06;GHS07 Danger Details