Online Database of Chemicals from Around the World
| Beijing Huafeng United Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.huafenginfo.com | |||
 | +86 (10) 5786-2036 57862181 65405760 +86 13366977697 | |||
 | +86 (10) 5786-2181 | |||
 | huafenginfo@126.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Discovery Fine Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.discofinechem.com | |||
 | +44 (1202) 874-517 | |||
 | +44 (845) 094-4385 | |||
 | pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Beijing Mesochem Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.mesochem.com | |||
 | +86 (10) 5786-2036 57862181 67374028 +86 13366977697 | |||
 | +86 (10) 5786-2181 | |||
 | sales@mesochem.com huafenginfo@126.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Hohance Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hohance.com | |||
 | +86 (21) 3111-5312 | |||
 | +86 (21) 3111-5317 | |||
 | info@hohance.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Selleck Chemicals LLC | USA | |||
|---|---|---|---|---|
 | www.selleckchem.com | |||
 | +1 (713) 535-9129 | |||
 | +1 (832) 582-8590 | |||
 | info@selleckchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Ningbo Easun Biological Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.easunbiotech.com | |||
 | +86 (574) 6208-9878 +86 18657120662 | |||
 | +86 (574) 6208-9877 | |||
 | sales@easunbiotech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Jinan Create Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jngcpharm.com | |||
 | +86 (531) 8806-5293 | |||
 | +86 (531) 8806-5293 | |||
 | 3166282224@qq.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Wuhan Haishan Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.haishankj.net | |||
 | +86 17720597715 | |||
 | +86 (27) 8663-7880 | |||
 | 3271849079@qq.com | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Classification | API >> Antineoplastic agents >> Antibiotic antineoplastic agents |
|---|---|
| Name | Epirubicin hydrochloride |
| Synonyms | (8S-cis)-10-[(3-Amino-2,3,6-trideoxy-alpha-L-arabino-hexopyranosyl)oxy]-7,8,9,10-tetrahydro-6,8,11-trihydroxy-8-(hydroxyacetyl)-1-methoxynaphthacene-5,12-dione hydrochloride |
| Molecular Structure | 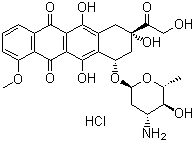 |
| Molecular Formula | C27H29NO11.HCl |
| Molecular Weight | 579.99 |
| CAS Registry Number | 56390-09-1 |
| EC Number | 260-145-2 |
| SMILES | C[C@H]1[C@@H]([C@H](C[C@@H](O1)O[C@H]2C[C@@](CC3=C2C(=C4C(=C3O)C(=O)C5=C(C4=O)C(=CC=C5)OC)O)(C(=O)CO)O)N)O.Cl |
| Solubility | 57 mg/mL (DMSO), <1 mg/mL (water), 57 mg/mL (ethanol) (Expl.) |
|---|---|
| Hazard Symbols |   GHS07;GHS08 Dander Details GHS07;GHS08 Dander Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H340-H350-H360 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P264-P270-P280-P301+P317-P318-P330-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Epirubicin hydrochloride is an anthracycline chemotherapy drug that was developed as a derivative of doxorubicin in the early 1980s. It was created by scientists at the pharmaceutical company Farmitalia Carlo Erba, with the goal of reducing the cardiotoxic effects of doxorubicin while maintaining its anticancer efficacy. Epirubicin is used to treat a variety of cancers, most notably breast cancer, where it plays a significant role in both adjuvant and neoadjuvant therapy. The chemical structure of epirubicin hydrochloride closely resembles that of doxorubicin, both of which are anthracyclines. The key difference is a structural modification of the hydroxyl group on the sugar moiety, which improves epirubicin's pharmacokinetics. This small change enhances the drug's efficacy and reduces the risk of cardiotoxicity, making it a more favorable option in some cases. Epirubicin works by intercalating into DNA, inhibiting the action of topoisomerase II, and preventing proper DNA replication and transcription. This process disrupts cell division, leading to apoptosis (cell death), particularly in rapidly dividing cancer cells. Epirubicin hydrochloride is most commonly used in the treatment of breast cancer. It has proven highly effective in treating early-stage breast cancer, especially when used as part of a combination chemotherapy regimen. Common regimens include FEC (5-fluorouracil, epirubicin, and cyclophosphamide), which has been a standard treatment for breast cancer for many years. Epirubicin is also used to treat other cancers, such as ovarian cancer, small cell lung cancer, and gastric cancer. Its use is generally as part of combination therapies, aiming to enhance the effectiveness of chemotherapy and prevent cancer recurrence. One of the key benefits of epirubicin over doxorubicin is its potentially lower risk of cardiotoxicity. Doxorubicin is known for causing heart damage, especially when administered in high doses or over long periods. Epirubicin, with its slightly altered structure, has a reduced likelihood of affecting heart function, though cardiotoxicity is still a concern. As with all anthracyclines, patients receiving epirubicin are monitored closely for signs of heart problems, and the drug is dosed carefully to minimize risks. The drug is also used in combination with other treatments, including radiation therapy. Studies have shown that epirubicin can enhance the effectiveness of radiation, which can be beneficial in certain clinical settings. Its ability to sensitize tumor cells to radiation makes it an attractive option in multi-modality cancer treatments. Despite its advantages, epirubicin has potential side effects. Common side effects include nausea, vomiting, hair loss, and bone marrow suppression, which can lead to reduced blood cell counts. This can increase the risk of infection, bleeding, and anemia, and requires close monitoring of blood counts during treatment. However, these side effects are often manageable with supportive care, such as the use of anti-nausea medications and drugs to stimulate blood cell production. Research continues to explore ways to improve the delivery and efficacy of epirubicin. Liposomal formulations of the drug are being developed to target tumors more effectively while reducing systemic toxicity. By encapsulating the drug in lipid nanoparticles, these formulations can increase drug concentration in tumor tissues and decrease exposure to healthy tissues, thus reducing side effects. In conclusion, epirubicin hydrochloride remains a cornerstone in the treatment of breast cancer and other cancers. Its ability to disrupt DNA replication in cancer cells while offering a potentially lower risk of cardiotoxicity has made it a valuable option in chemotherapy regimens. Though it is associated with common chemotherapy side effects, the overall benefits in cancer treatment continue to make it an important therapeutic tool. Ongoing research into formulations and combination therapies may further enhance its clinical applications in oncology. References 2024. Exploring Radioiodinated Anastrozole and Epirubicin as AKT1-Targeted Radiopharmaceuticals in Breast Cancer: In Silico Analysis and Potential Therapeutic Effect with Functional Nuclear Imagining Implications. Molecules (Basel, Switzerland), 29(17). DOI: 10.3390/molecules29174203 1991. A prospective randomized trial comparing epirubicin monochemotherapy to two fluorouracil, cyclophosphamide, and epirubicin regimens differing in epirubicin dose in advanced breast cancer patients. The French Epirubicin Study Group. Journal of clinical oncology : official journal of the American Society of Clinical Oncology, 9(2). DOI: 10.1200/jco.1991.9.2.305 1987. Metabolism of epidoxorubicin in animals: absence of glucuronidation. Cancer Chemotherapy and Pharmacology, 20(2). DOI: 10.1007/bf00252968 |
| Market Analysis Reports |