Online Database of Chemicals from Around the World
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Smartchem (Beijing) Ltd. | China | |||
|---|---|---|---|---|
 | www.smartchembeijing.com | |||
 | +86 (10) 5720-3829 5130-5129 +86 13301124250 | |||
 | +86 (10) 5130-5388 | |||
 | sales@smartchembeijing.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| RIA International LLC | USA | |||
|---|---|---|---|---|
 | www.riausa.net | |||
 | +1 (973) 581-1282 | |||
 | +1 (973) 581-1283 | |||
 | marketing@riausa.net ria@riausa.net | |||
| Chemical distributor since 1986 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Angewchem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.angewchem.com | |||
 | +86 18917157435 | |||
 | dylan@angewchem.com | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Pure Synmr Ingredients Limited | China | |||
|---|---|---|---|---|
 | www.synmr.com | |||
 | +86 (21) 6045-7587 ex 806 | |||
 | sven@puresynmr.com | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Xi'an Yinherb Bio-tech Co., Ltd | China | |||
|---|---|---|---|---|
 | yinherb.ecer.com | |||
 | +86 (29) 6333-2330 | |||
 | sales@yinherb-nutraceuticals.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Natural product >> Steroidal compound |
|---|---|
| Name | (3beta,5alpha,25R)-3,5-Dihydroxyspirostan-6-one |
| Synonyms | BB 16; Biobras 16; Brassinosteroid BB 16; DI 31 |
| Molecular Structure | 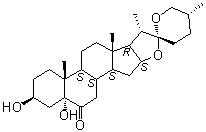 |
| Molecular Formula | C27H42O5 |
| Molecular Weight | 446.62 |
| CAS Registry Number | 56786-63-1 |
| EC Number | 884-776-3 |
| SMILES | C[C@@H]1CC[C@@]2([C@H]([C@H]3[C@@H](O2)C[C@@H]4[C@@]3(CC[C@H]5[C@H]4CC(=O)[C@@]6([C@@]5(CC[C@@H](C6)O)C)O)C)C)OC1 |
| Solubility | Practically insoluble (0.025 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.21±0.1 g/cm3 (20 °C 760 Torr), Calc.* |
| Melting point | 268-270 °C (ethanol )** |
| Boiling point | 578.4±50.0 °C 760 mmHg (Calc.)* |
| Flash point | 188.5±23.6 °C (Calc.)* |
| Index of refraction | 1.573 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2015 ACD/Labs) |
| ** | Rivera, Daniel G. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details |
|
(3beta,5alpha,25R)-3,5-Dihydroxyspirostan-6-one is a naturally occurring steroidal sapogenin classified within the spirostanol group of compounds. It is structurally characterized by a spiroketal system involving a spiro junction at the C-22 and C-26 positions, with hydroxyl groups at the C-3 and C-5 positions and a ketone functional group at the C-6 position. This compound belongs to a broader class of steroidal sapogenins that are mainly found in plant species from the Dioscoreaceae, Liliaceae, and Asparagaceae families, and are frequently studied for their pharmacological and biochemical properties. The discovery of (3beta,5alpha,25R)-3,5-dihydroxyspirostan-6-one traces back to investigations into traditional medicinal plants, particularly those used in Chinese, Ayurvedic, and Latin American ethnomedicine. The sapogenin core of this compound is often found as the aglycone component in saponins, which are glycosidic natural products exhibiting surfactant properties. Early phytochemical explorations into Dioscorea species—commonly known as yams—revealed a wide array of spirostanol and furostanol derivatives. Among these, derivatives of diosgenin and related sapogenins drew significant interest due to their ability to serve as precursors for the semi-synthesis of steroid hormones. This compound exhibits a stereospecific arrangement, notably with the 5alpha hydrogen configuration and the 25R chiral center, which contribute to its biological activity. While it may be present in minor amounts in natural sources compared to more abundant sapogenins like diosgenin or hecogenin, (3beta,5alpha,25R)-3,5-dihydroxyspirostan-6-one can be isolated via hydrolysis of its saponin precursors followed by chromatographic techniques. In terms of applications, the compound and its analogues are of interest in both the pharmaceutical and biochemical research sectors. Structurally similar sapogenins are used as intermediates for the industrial production of corticosteroids, sex hormones (including progesterone and testosterone), and oral contraceptives. The spirostan skeleton also provides a unique framework for the synthesis of bioactive molecules with potential antifungal, anti-inflammatory, and antitumor properties. Pharmacologically, spirostanol-type compounds have been evaluated for their interaction with cell membranes and enzymes, especially due to their amphiphilic nature. Although (3beta,5alpha,25R)-3,5-dihydroxyspirostan-6-one itself has not been widely commercialized or used directly in therapeutic agents, it has been included in bioactivity screening studies for potential cytotoxic and antiprotozoal properties. Additionally, sapogenins of this nature are explored for their adjuvant effects in vaccine formulations and as drug delivery agents, owing to their ability to form micelles and permeabilize biological membranes. Modern research has continued to explore spirostanol compounds using techniques such as nuclear magnetic resonance (NMR), mass spectrometry, and X-ray crystallography for structural elucidation, as well as computational docking to assess potential molecular targets. The biosynthetic pathways of such compounds in plants are also being investigated, particularly the enzymatic transformations that yield specific stereochemical configurations at the C-5 and C-25 positions. In the context of chemotaxonomy, the presence of (3beta,5alpha,25R)-3,5-dihydroxyspirostan-6-one and its derivatives can serve as biomarkers to differentiate plant species within the same genus or family. This aids in the quality control and standardization of plant-derived medicinal products, especially those marketed in the herbal supplement industry. Overall, (3beta,5alpha,25R)-3,5-dihydroxyspirostan-6-one stands as a minor but significant member of the spirostanol sapogenin group, with ongoing interest in its pharmacological potential, structural uniqueness, and role as a precursor in steroid biosynthesis pathways. References 2021. Brassinosteroid and brassinosteroid-mimic differentially modulate Arabidopsis thaliana fitness under drought. Plant Growth Regulation, 95(1). DOI: 10.1007/s10725-021-00722-8 |
| Market Analysis Reports |