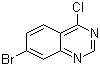
7-Bromo-4-chloroquinazoline is a halogenated quinazoline derivative with potential applications in various fields, including medicinal chemistry and materials science. Quinazoline is a bicyclic compound containing a benzene ring fused to a pyrimidine ring, which can be functionalized at different positions to alter its chemical and biological properties. In this case, the compound features bromine at the 7-position and chlorine at the 4-position of the quinazoline ring.
The synthesis of 7-bromo-4-chloroquinazoline generally involves halogenation of a quinazoline precursor. The introduction of the bromine and chlorine atoms is typically carried out through electrophilic aromatic substitution reactions. These halogenation reactions are selective, with the bromine atom typically being introduced at the 7-position, and the chlorine atom introduced at the 4-position, due to the electron-withdrawing nature of the pyrimidine ring, which directs the halogenation to these positions.
In medicinal chemistry, quinazoline derivatives are of interest due to their biological activities. Quinazoline-based compounds have been found to possess a variety of pharmacological properties, including anticancer, antimicrobial, and anti-inflammatory activities. The presence of halogen atoms, such as bromine and chlorine, can further enhance these properties, particularly by modifying the lipophilicity, reactivity, and molecular interactions with biological targets. The halogens can also influence the molecule’s ability to penetrate biological membranes, which is an important consideration for drug design.
For example, quinazoline derivatives, including halogenated compounds like 7-bromo-4-chloroquinazoline, have been studied for their ability to inhibit specific kinases, which are enzymes involved in cell signaling pathways. Kinase inhibitors are important in the treatment of various diseases, particularly cancer, where abnormal kinase activity often leads to uncontrolled cell growth. The presence of the halogens in 7-bromo-4-chloroquinazoline could contribute to its potential as a kinase inhibitor or as part of a more complex drug development strategy targeting specific molecular pathways.
Beyond pharmaceuticals, 7-bromo-4-chloroquinazoline may have applications in the development of functional materials. Quinazoline derivatives, particularly those with halogen substitutions, can exhibit interesting electronic and optical properties. They may be utilized in the synthesis of advanced materials, such as conducting polymers, organic semiconductors, or fluorescent probes, due to their ability to interact with light or electric fields in specific ways. The halogen atoms may also be used to fine-tune the molecular properties of quinazoline-based materials for use in sensors or other electronic devices.
The chemical reactivity of 7-bromo-4-chloroquinazoline also makes it a useful intermediate in organic synthesis. The halogens on the quinazoline ring provide sites for further functionalization, allowing the compound to be transformed into a wide range of derivatives. This versatility is beneficial in both the synthesis of new chemical entities for biological testing and the development of new materials with specific properties.
In summary, 7-bromo-4-chloroquinazoline is a halogenated quinazoline derivative with diverse applications in medicinal chemistry, materials science, and organic synthesis. Its potential as a pharmacologically active compound, particularly as a kinase inhibitor, as well as its usefulness as a building block for more complex molecules, makes it a valuable subject of ongoing research. The presence of halogens adds to its chemical versatility, enabling it to be further modified for various scientific and industrial purposes.
|


























 GHS06;GHS07 Danger Details
GHS06;GHS07 Danger Details