Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| ShangHai PuYi Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.gyrochem.com | |||
 | +86 (21) 5768-7505 ex 305 +86 13601674297 +86 15601977133 | |||
 | +86 (21) 5768-7512 | |||
 | sales@gyrochem.com annafan@gyrochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Sinfachem Limited | China | |||
|---|---|---|---|---|
 | www.sinfachem.com | |||
 | +86 (25) 8468-3399 8517-8591 | |||
 | +86 (25) 8468-3112 | |||
 | sinfachem@foxmail.com sinfachemltd@foxmail.com info@sinfachem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Sino-Future Bio-Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sino-future.com | |||
 | +86 (29) 8318-2005 | |||
 | sinofuture@vip.163.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Nanning Weikang Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.weikangpharm.com | |||
 | +86 (771) 451-6110 | |||
 | +86 (771) 451-6130 | |||
 | sales@weikangpharm.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Rosewa (HK) Holding Group Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rosewachem.com | |||
 | +86 18983840262 | |||
 | +86 (23) 6283-3877 | |||
 | sales@rosewachem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Creative Peptides | USA | |||
|---|---|---|---|---|
 | www.creative-peptides.com | |||
 | +1 (631) 624-4882 | |||
 | +1 (631) 614-7828 | |||
 | info@creative-peptides.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Guangzhou Ku Wei Biological Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kuvlle.com | |||
 | +86 (20) 6106-3208 | |||
 | +86 (20) 6106-3208 | |||
 | yxusmith@aliyun.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Aktin Chemicals Inc | China | |||
|---|---|---|---|---|
 | www.aktinchem.com | |||
 | +86 400-028-7725 | |||
 | +86 (28) 8515-2372 | |||
 | info@aktinchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Intatrade Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.intatrade.de | |||
 | +49 (3493) 605-465 | |||
 | +49 (3493) 605-470 | |||
 | sales@intatrade.de | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Forward Farma Inc | USA | |||
|---|---|---|---|---|
 | www.forwardfarma.com | |||
 | +1 (909) 963-1481 | |||
 | xiaye@forwardfarma.com | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Chengdu Biopurify Phytochemicals Ltd. | China | |||
|---|---|---|---|---|
 | www.phytopurify.com | |||
 | +86 (28) 8263-3860 8263-3987 | |||
 | +86 (28) 8263-3165 | |||
 | sales@biopurify.com biopurify@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2017 | ||||
| Hangzhou Cyanochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cyanochem.com | |||
 | +86 (571) 8522-0831 +86 17788583750 | |||
 | +86 (571) 8522-0831 | |||
 | info@cyanochem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Taizhou Bona Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tzbnchem.com | |||
 | +86 (576) 8831-2366 | |||
 | +86 (576) 8870-7069 | |||
 | kitty@tzbnchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Shanghai Sunflower Pharmachem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sunflower-chem.com | |||
 | +86 (021) 866-7323 | |||
 | 1944991209@qq.com | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Wuhan Yuancheng Technology Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.yuanchengtechnology.com | |||
 | +86 17282536078 | |||
 | +86 17282536078 | |||
 | 3042184429@qq.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: 17282536078 | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Sinova Corporation | USA | |||
|---|---|---|---|---|
 | www.sinovainc.com | |||
 | +1 (301) 961-1525 | |||
 | +1 (240) 235-4288 | |||
 | sales@sinovainc.com | |||
| Chemical manufacturer | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| Watanabe Chemical Ind., Ltd. | Japan | |||
|---|---|---|---|---|
 | www.watanabechem.co.jp | |||
 | +81 (82) 231-0540 | |||
 | +81 (82) 231-1451 | |||
 | inquiry@watanabechem.co.jp | |||
| Chemical manufacturer | ||||
| Cayman Chemical Company | USA | |||
|---|---|---|---|---|
 | www.caymanchem.com | |||
 | +1 (734) 971-3335 | |||
 | +1 (734) 971-3640 | |||
 | sales@caymanchem.com | |||
| Chemical manufacturer | ||||
| Acros Organics | Belgium | |||
|---|---|---|---|---|
 | www.acros.com | |||
 | +86 (21) 5258-1100 | |||
 | +86 (21) 5258-0119 | |||
 | info@acros.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Metal halides and halides >> Metal chlorides and salts |
|---|---|
| Name | Levodopa |
| Synonyms | 3-(3,4-Dihydroxyphenyl)-L-alanine; L-3-(3,4-Dihydroxyphenyl)alanine; L-DOPA |
| Molecular Structure | 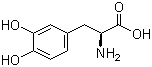 |
| Molecular Formula | C9H11NO4 |
| Molecular Weight | 197.19 |
| Protein Sequence | X |
| CAS Registry Number | 59-92-7 |
| EC Number | 200-445-2 |
| SMILES | C1=CC(=C(C=C1C[C@@H](C(=O)O)N)O)O |
| Solubility | Soluble 5 mM (water) |
|---|---|
| Melting point | 295 °C |
| alpha | -11.7 ° (c=5.3, 1N HCl) |
| Hazard Symbols |    GHS07;GHS08;GHS09 Danger Details GHS07;GHS08;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335-H361d-H372-H411-H412 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P260-P261-P264-P264+P265-P270-P271-P273-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P318-P319-P321-P330-P332+P317-P337+P317-P362+P364-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Levodopa, also known as L-DOPA, is a naturally occurring chemical precursor to dopamine, an essential neurotransmitter in the brain. Its discovery dates back to the early 20th century, when it was first isolated from the seeds of the broad bean plant, *Vicia faba*. In 1913, Levodopa was synthesized for the first time by the Polish biochemist Casimir Funk, but its potential therapeutic applications were not realized until much later. During the 1960s, Levodopa gained attention as a treatment for Parkinson’s disease, thanks to the pioneering work of neurologists George Cotzias and Oleh Hornykiewicz. They demonstrated that administering Levodopa could replenish the brain's dopamine levels, offering significant symptom relief to Parkinson's patients. Levodopa is converted into dopamine by the enzyme aromatic L-amino acid decarboxylase (AAAD) once it crosses the blood-brain barrier. Dopamine itself cannot cross this barrier, making Levodopa an essential prodrug for increasing dopamine concentrations in the brain. The discovery of this mechanism revolutionized the treatment of Parkinson’s disease, a neurodegenerative disorder characterized by the loss of dopamine-producing neurons. Parkinson's patients typically experience tremors, rigidity, and bradykinesia, all of which can be alleviated by Levodopa therapy, making it the gold standard for treatment. Levodopa is commonly administered in combination with carbidopa, a peripheral decarboxylase inhibitor that prevents the premature conversion of Levodopa into dopamine outside the brain, thereby increasing its efficacy and reducing side effects such as nausea and cardiovascular complications. Beyond Parkinson’s disease, Levodopa has been explored in the treatment of other movement disorders, including Restless Leg Syndrome (RLS) and dystonia. Its ability to restore dopamine levels has been beneficial in managing symptoms of these disorders as well. Furthermore, Levodopa has been studied in the context of certain neuropsychiatric conditions, although its primary therapeutic use remains in the management of Parkinson’s disease. In addition to its medical applications, Levodopa’s role as a precursor in the biosynthesis of catecholamines has made it a valuable tool in neurobiological research. It has been used to investigate dopamine pathways and the broader understanding of neurotransmitter systems in both health and disease. Research on Levodopa has contributed significantly to the understanding of neurodegenerative disorders and the development of new therapeutic approaches that target dopamine signaling. The limitations of Levodopa therapy have also been a focus of research, particularly concerning its long-term use. Chronic administration can lead to motor complications such as dyskinesia (involuntary movements) and fluctuations in drug efficacy, known as the "on-off" phenomenon. Despite these challenges, Levodopa remains a cornerstone of Parkinson’s treatment, and efforts continue to optimize its use, including exploring alternative formulations like controlled-release versions and combination therapies to improve patient outcomes. References 1979. Behavioral study on the interactions between trazodone and L-dopa. Pharmacological Research Communications, 11(3). DOI: 10.1016/s0031-6989(79)80088-0 1979. Levodopa and dopamine analogs: melanin precursors as antitumor agents in experimental human and murine leukemia. Cancer Treatment Reports, 63(6). URL: 466656 1979. Pharmacokinetics of bromocriptine during continuous oral treatment of Parkinson's disease. European Journal of Clinical Pharmacology, 15(4). DOI: 10.1007/bf00618517 |
| Market Analysis Reports |