Online Database of Chemicals from Around the World
| Zhejiang Sixian Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sx-pharm.com | |||
 | +86 (575) 8557-5861 8557-5010 8557-4620 | |||
 | +86 (575) 8557-5010 | |||
 | sxpharm@vip.163.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Tianjin Zhongxin Chem-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tjzxchem.com | |||
 | +86 (22) 6688-0623 | |||
 | +86 (22) 8988-0739 ex 8030 | |||
 | sales@tjzxchem.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Huaian Yishu Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hayishu.com | |||
 | +86 (517) 8588-3738 | |||
 | +86 (517) 8588-3738 | |||
 | 8998145@qq.com | |||
| Chemical manufacturer since 2018 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Shineme Chemfar Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemfar.com | |||
 | +86 (731) 8471-8061 | |||
 | +86 (731) 8471-6269 | |||
 | sales@chemfar.com | |||
| Chemical manufacturer since 1996 | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives >> Cyclic carboxylic acid |
|---|---|
| Name | 1,3-Bisbenzyl-2-oxoimidazolidine-4,5-dicarboxylic acid |
| Synonyms | 1,3-dibenzyl-2-oxoimidazolidine-4,5-dicarboxylic acid |
| Molecular Structure | 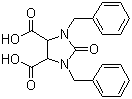 |
| Molecular Formula | C19H18N2O5 |
| Molecular Weight | 354.36 |
| CAS Registry Number | 59564-78-2 |
| EC Number | 261-806-8 |
| SMILES | C1=CC=C(C=C1)CN2C(C(N(C2=O)CC3=CC=CC=C3)C(=O)O)C(=O)O |
| Density | 1.4±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 170-174 °C |
| Index of Refraction | 1.659, Calc.* |
| Boiling Point | 654.2±55.0 °C (760 mmHg), Calc.* |
| Flash Point | 349.5±31.5 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319 Details |
| Safety Statements | P264-P264+P265-P280-P302+P352-P305+P351+P338-P321-P332+P317-P337+P317-P362+P364 Details |
| SDS | Available |
|
1,3-Bisbenzyl-2-oxoimidazolidine-4,5-dicarboxylic acid is a heterocyclic compound that has attracted interest in the fields of organic synthesis and medicinal chemistry due to its unique structural properties and potential applications. This compound is characterized by its imidazolidine ring, which is fused with two benzyl groups and carries two carboxylic acid functionalities. The compound's discovery can be traced back to ongoing research in the mid-20th century that aimed to explore the synthesis of imidazolidine derivatives and their corresponding biological activities. The synthesis of 1,3-bisbenzyl-2-oxoimidazolidine-4,5-dicarboxylic acid involves a multi-step process, typically beginning with the formation of the imidazolidine ring from appropriate precursors such as α-amino acids or α-keto acids. The reaction conditions, including temperature, solvents, and catalysts, play a critical role in obtaining the desired yield and purity of the product. Over the years, various synthetic routes have been developed to optimize this process, leading to an increased understanding of the compound's chemical behavior and stability. One of the most significant applications of 1,3-bisbenzyl-2-oxoimidazolidine-4,5-dicarboxylic acid lies in its potential as a bioactive agent. Research has shown that this compound exhibits promising antimicrobial and anti-inflammatory properties, making it a candidate for the development of new pharmaceuticals. In vitro studies have demonstrated its effectiveness against various bacterial strains, suggesting that it may serve as a lead compound for antibiotic development. The compound's ability to modulate inflammatory pathways also presents opportunities for treating chronic inflammatory diseases. In addition to its biological applications, 1,3-bisbenzyl-2-oxoimidazolidine-4,5-dicarboxylic acid is being explored for its use in the field of materials science. Its unique chemical structure allows for the potential development of novel polymers and resins with enhanced thermal and mechanical properties. Researchers are investigating its utility as a crosslinking agent or as part of composite materials, particularly in applications requiring lightweight and durable components. The compound's ability to act as a chelating agent has garnered attention in coordination chemistry as well. Its multiple carboxylic acid groups can coordinate with metal ions, forming stable complexes that may find applications in catalysis or metal recovery processes. These properties enhance its versatility as a chemical building block in the synthesis of more complex structures. In conclusion, 1,3-bisbenzyl-2-oxoimidazolidine-4,5-dicarboxylic acid is a compound of significant interest due to its unique structural features and diverse range of applications. Its discovery has paved the way for ongoing research into its biological activity and potential industrial uses. As scientists continue to explore its properties and reactivity, this compound may lead to the development of novel therapeutic agents and advanced materials that could benefit various sectors, including healthcare and manufacturing. References 2005. Synthetic Studies on d-Biotin, Part 9. An Improved Asymmetric Synthetic Route to d-Biotin via Hoffmann-Roche Lactone-Thiolactone Approach. Chemical and Pharmaceutical Bulletin, 53(7). DOI: 10.1248/cpb.53.743 2022. Structures and Magnetic Properties of Binuclear Co(II) and Dy(III) Complexes with Cis-1,3-Dibenzyl-2-Imidazolidone-4,5-Dicarboxylic Acid of Variable Conformations. Journal of Cluster Science, 34(2). DOI: 10.1007/s10876-022-02254-1 2018. New Zn/Cd Coordination Polymers Constructed from Mixed Ligands: Crystal Structures and Photocatalytic Performances Toward Organic Dyes Degradation. Journal of Inorganic and Organometallic Polymers and Materials, 28(3). DOI: 10.1007/s10904-018-0804-0 |
| Market Analysis Reports |