Online Database of Chemicals from Around the World
| Extrasynthese Chemical S.A.S. | France | |||
|---|---|---|---|---|
 | www.extrasynthese.com | |||
 | +33 (47) 898-2034 | |||
 | +33 (47) 898-1945 | |||
 | info@extrasynthese.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Shanghai Kewel Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kewelchem.com | |||
 | +86 (21) 6460-9169 | |||
 | +86 (21) 6460-9160 | |||
 | kewelchem88@gmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Nanjing Rich Native Animal Products Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.njrich.com | |||
 | +86 (25) 8369-0347 | |||
 | +86 (25) 8369-4952 | |||
 | sales@njrich.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Witofly Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.witofly.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales@witofly.com | |||
 | QQ Chat | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shandong Zhishang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zhishangchemical.com | |||
 | +86 17653113209 13065062079 | |||
 | sales001@sdzschem.com sales002@sdzschem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Shandong Look Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sdlookchem.com | |||
 | +86 17653113219 | |||
 | sales01@sdlookchem.com | |||
 | QQ Chat | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Riverland Trading LLC. | USA | |||
|---|---|---|---|---|
 | www.riverlandtrading.com | |||
 | +1 (336).944-2293 | |||
 | bens@riverlandtrading.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2023 | ||||
| Jiangxi Zhonghuan New Materials Co., Ltd | China | |||
|---|---|---|---|---|
 | www.jxzhxcl.com | |||
 | 13766287383 | |||
 | 2246992030@qq.com | |||
 | QQ Chat | |||
 | WeChat: 13766287383 | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Jiangxi Xinsen Natural Vegetable Oil Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jxxinsen.com | |||
 | +86 18679667531 | |||
 | 592432494@qq.com | |||
 | QQ Chat | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Ji'an Guoguang Fragrance Factory | China | |||
|---|---|---|---|---|
 | www.jxggxlc.com | |||
 | +86 18607064978 | |||
 | 2529415945@qq.com | |||
 | QQ Chat | |||
| Chemical distributor since 1988 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| Takasago International Corp. (USA) | USA | |||
|---|---|---|---|---|
 | www.takasago.com | |||
 | +1 (201) 767-9001 | |||
 | +1 (201) 784-7277 | |||
 | aromachemicals@takasago.com | |||
| Chemical manufacturer | ||||
| ChemPur GmbH | Germany | |||
|---|---|---|---|---|
 | www.chempur.de | |||
 | +49 (721) 933-840 | |||
 | +49 (721) 47-2001 | |||
 | info@chempur.de | |||
| Chemical manufacturer | ||||
| Destilaciones Bordas Chinchurreta SA | Spain | |||
|---|---|---|---|---|
 | www.bordas-sa.com | |||
 | +34 (95) 441-9000 | |||
 | +34 (95) 441-7152 / 453-1229 | |||
 | bordas@bordas-sa.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Olefins (cyclic and non-cyclic) |
|---|---|
| Name | (R)-(+)-Limonene |
| Synonyms | (R)-4-Isopropenyl-1-methylcyclohexene; (+)-p-Mentha-1,8-diene; (+)-Carvene |
| Molecular Structure | 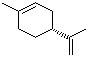 |
| Molecular Formula | C10H16 |
| Molecular Weight | 136.23 |
| CAS Registry Number | 5989-27-5 |
| EC Number | 227-813-5 |
| FEMA | 2633 |
| SMILES | CC1=CC[C@@H](CC1)C(=C)C |
| Density | 0.842 |
|---|---|
| Melting point | -74 °C |
| Boiling point | 176-177 °C |
| Refractive index | 1.473 |
| Flash point | 48 °C |
| alpha | 112 ° (10% in ethanol) |
| Hazard Symbols |     GHS02;GHS07;GHS08;GHS09 Warning Details GHS02;GHS07;GHS08;GHS09 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226-H304-H315-H317-H319-H400-H410 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P261-P264-P264+P265-P272-P273-P280-P301+P316-P302+P352-P303+P361+P353-P305+P351+P338-P321-P331-P332+P317-P333+P317-P337+P317-P362+P364-P370+P378-P391-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2052 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|
(R)-(+)-limonene, often referred to simply as limonene, was first isolated from citrus oils by German chemist Otto Wallach in the early 19th century. It is abundant in the essential oils of citrus fruits such as oranges and lemons and became the focus of early natural product research. Limonene exists in two isomeric forms, but the (R)-(+)-enantiomer has a characteristic orange aroma and is the one most commonly found in nature. Limonene is a cyclic monoterpene with the molecular formula C10H16. The (R)-(+)-enantiomer is a clear, colorless liquid at room temperature with high volatility and a distinctive citrus aroma. Its hydrophobicity and compatibility with a wide range of organic solvents enhance its usefulness in numerous formulations. In the food and beverage industry, (R)-(+)-limonene is used as a flavoring agent. Its fresh, tangy aroma enhances the taste of beverages, confectionery, and baked goods. Recognized as generally safe by regulatory agencies such as the FDA, limonene is a natural alternative to synthetic fragrances, in line with consumer preferences for clean label ingredients. The attractive scent of (R)-(+)-limonene makes it a popular ingredient in perfumes, colognes, and personal care products. Its volatility allows it to be used as a top note in perfumes, bringing a fresh style that captures the essence of citrus. In addition, it is used in lotions, shampoos, and cleaning products to give off a pleasant scent that helps increase product appeal. In medicine, (R)-(+)-limonene is promising for its anti-inflammatory and antioxidant properties. It is used in topical formulations and as a potential therapeutic agent in various studies. Its action as a solvent enhances the delivery of active ingredients in pharmaceutical formulations and cosmetics. In industry, (R)-(+)-limonene is used as a green solvent in cleaning products and paints due to its effectiveness in dissolving oils and fats. Its low toxicity and biodegradability make it an attractive option for environmentally friendly solutions. In addition, it is used to synthesize biodegradable plastics and as a bio-based feedstock in green chemistry. While (R)-(+)-limonene is generally regarded as safe, it can cause skin irritation in sensitive individuals and should be handled with care in concentrated forms. Proper labeling and usage guidelines can help reduce potential risks and ensure safe consumption. As a natural compound, (R)-(+)-limonene is consistent with sustainable development initiatives. It is obtained from citrus byproducts, promoting waste utilization and reducing the environmental footprint of the citrus processing industry. Its biodegradability further supports its role in creating environmentally friendly products and processes. Ongoing research aims to expand the application of (R)-(+)-limonene in emerging fields such as biotechnology and renewable materials. Advances in extraction and synthesis technologies are expected to increase its availability and purity, promoting new uses in different fields. References 2025. Biphasic effect of limonene on contraction of isolated rat aorta. Chemico-Biological Interactions, 409. DOI: 10.1016/j.cbi.2024.111313 2025. Efficient epoxidation of (R)-(+)-limonene to limonene dioxide through peracids generated using whole-cell Rhizopus oryzae lipase. Bioresource Technology, 417. DOI: 10.1016/j.biortech.2024.131645 2024. Limonene oxide isomerization: a broad study of acid catalyst effects. Reaction Kinetics, Mechanisms and Catalysis, 137(6). DOI: 10.1007/s11144-024-02774-z |
| Market Analysis Reports |