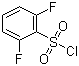
2,6-Difluorobenzenesulfonyl chloride is a functionalized aromatic sulfonyl halide in which a benzene ring is substituted with fluorine atoms at the 2- and 6-positions and a sulfonyl chloride group at the 1-position. Sulfonyl chlorides are highly reactive electrophilic compounds widely used in organic synthesis for the preparation of sulfonamides, sulfonate esters, and other derivatives. The presence of the fluorine atoms influences the electronic properties of the aromatic ring, increasing its electron-withdrawing character, which in turn enhances the electrophilicity of the sulfonyl chloride and affects reactivity in nucleophilic substitution reactions.
Structurally, the molecule consists of a benzene ring with two fluorine atoms positioned ortho to the sulfonyl chloride group. The sulfonyl chloride functional group (–SO2Cl) contains a sulfur atom doubly bonded to two oxygen atoms and bonded to a chlorine atom, making it a strong electrophile capable of reacting with nucleophiles such as amines, alcohols, or thiols to form sulfonamides, sulfonates, or thioesters. The ortho-fluorine substituents add steric hindrance and electronic effects that can influence reaction selectivity and the stability of intermediates.
The synthesis of 2,6-difluorobenzenesulfonyl chloride is typically achieved through the chlorosulfonation of 1,3-difluorobenzene using chlorosulfonic acid or sulfuryl chloride under controlled conditions. The fluorine substituents direct the sulfonylation to the 1-position due to electronic and steric effects, allowing selective formation of the desired product. The resulting sulfonyl chloride is a stable yet highly reactive intermediate suitable for further chemical transformations.
In organic and medicinal chemistry, 2,6-difluorobenzenesulfonyl chloride is used as a key intermediate for synthesizing sulfonamide-based compounds, which are important in pharmaceuticals, agrochemicals, and functional materials. The ortho-fluorine atoms can enhance the metabolic stability, lipophilicity, and binding affinity of sulfonamide derivatives by influencing electronic density and steric properties. Reactions with amines lead to sulfonamide linkages, which are widely present in antibacterial agents, enzyme inhibitors, and receptor ligands.
Beyond medicinal chemistry, this compound serves as a versatile building block in organic synthesis. The reactive sulfonyl chloride allows the formation of sulfonate esters and other functionalized derivatives. The electron-withdrawing fluorine substituents can also influence the acidity of adjacent hydrogen atoms and the reactivity of the aromatic ring, enabling further selective functionalizations such as electrophilic or nucleophilic aromatic substitutions. This combination of reactivity and tunable electronic properties makes the compound valuable for the preparation of advanced intermediates and functional materials.
Overall, 2,6-difluorobenzenesulfonyl chloride is a reactive and versatile aromatic sulfonyl chloride featuring ortho-fluorine substituents. Its chemical properties and structural features make it a valuable intermediate for the synthesis of sulfonamides, sulfonate esters, and other functional derivatives, with applications in medicinal chemistry, organic synthesis, and materials science.
References
2017. Synthesis, anti-angiogenic and DNA cleavage studies of novel N-(4-methyl-3-((4-(pyridin-3-yl)pyrimidin-2-yl)amino)phenyl)piperidine-4-carboxamide derivatives. Chemistry Central Journal.
DOI: 10.1186/s13065-017-0354-5
|



















































 GHS05 Danger Details
GHS05 Danger Details