Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Discovery Fine Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.discofinechem.com | |||
 | +44 (1202) 874-517 | |||
 | +44 (845) 094-4385 | |||
 | pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Nile Chemicals | India | |||
|---|---|---|---|---|
 | www.nilechemicals.com | |||
 | +91 (22) 6631-3162 | |||
 | +91 (22) 2205-4833 | |||
 | sales@nilechemicals.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Shanghai Luyuan Fine Chemical Factory | China | |||
|---|---|---|---|---|
 | www.finechem-china.com | |||
 | +86 (21) 6998-8805 6998-8373 5952-2390 | |||
 | +86 (21) 6998-8155 | |||
 | huatingchem@gmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Tianjin Zhongxin Chem-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tjzxchem.com | |||
 | +86 (22) 6688-0623 | |||
 | +86 (22) 8988-0739 ex 8030 | |||
 | sales@tjzxchem.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| JIANGSU KOLOD FOOD INGREDIENTS CO.,LTD. | China | |||
|---|---|---|---|---|
 | www.kolod.com | |||
 | +86 18762571399 | |||
 | +86 (518) 8511-5307 | |||
 | sales1399@jskolod.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2020 | ||||
| J&H Chemical HK Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jhechem.com | |||
 | +86 (571) 8739-6430 8739-6432 | |||
 | +86 (571) 8739-6431 | |||
 | sales@jhechem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| Celtic Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.celticchemicals.co.uk | |||
 | +44 (1656) 749-358 | |||
 | +44 (1656) 746-490 | |||
 | sales@celticchemicals.co.uk | |||
| Chemical manufacturer | ||||
| Materion | USA | |||
|---|---|---|---|---|
 | www.materion.com | |||
 | +1 (216) 486-4200 | |||
 | +1 (414) 289-9805 | |||
 | info@materion.com | |||
| Chemical manufacturer | ||||
| Gelest, Inc. | USA | |||
|---|---|---|---|---|
 | www.gelest.com | |||
 | +1 (215) 547-1015 | |||
 | +1 (215) 547-2484 | |||
 | info@gelest.com | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| World Metal, LLC | USA | |||
|---|---|---|---|---|
 | www.worldmetalllc.com | |||
 | +1 (877) 363-8257 (281) 491-7474 | |||
 | +1 (281) 491-7979 | |||
 | sales@worldmetalllc.com | |||
| Chemical manufacturer | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| ChemPur GmbH | Germany | |||
|---|---|---|---|---|
 | www.chempur.de | |||
 | +49 (721) 933-840 | |||
 | +49 (721) 47-2001 | |||
 | info@chempur.de | |||
| Chemical manufacturer | ||||
| ProChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.prochemonline.com | |||
 | +1 (815) 398-1788 | |||
 | +1 (815) 398-1810 | |||
 | prochem3@aol.com | |||
| Chemical manufacturer since 1986 | ||||
| Classification | Organic raw materials >> Organometallic salt |
|---|---|
| Name | Cupric acetate monohydrate |
| Synonyms | Acetic acid copper salt monohydrate; Copper(II) acetate monohydrate |
| Molecular Structure | 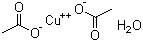 |
| Molecular Formula | Cu(C2H3O2)2.H2O |
| Molecular Weight | 199.65 |
| CAS Registry Number | 6046-93-1 |
| EC Number | 611-978-9 |
| SMILES | CC(=O)[O-].CC(=O)[O-].O.[Cu+2] |
| Density | 1.882 |
|---|---|
| Melting point | 115 °C (dec.) |
| Boiling point | 240 °C |
| Water solubility | 72 g/L (20 °C) |
| Hazard Symbols |    GHS05;GHS07;GHS09 Danger Details GHS05;GHS07;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H314-H315-H318-H319-H335-H400-H410-H411 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P261-P264-P264+P265-P270-P271-P273-P280-P301+P317-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P351+P338-P305+P354+P338-P316-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P363-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 9106; UN 3077 | ||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||
|
Cupric acetate monohydrate, with the chemical formula Cu(C2H3O2)2·H2O, is a blue crystalline compound that plays a vital role in various chemical processes and applications. This compound is a hydrate of cupric acetate, consisting of copper ions combined with acetate anions and water molecules. Its discovery dates back to the early exploration of copper compounds, with documented uses in both the arts and sciences as far back as the 18th century. The compound's unique properties and versatility have led to its widespread application across several industries. One of the primary applications of cupric acetate monohydrate is in organic synthesis. It serves as a key reagent in various chemical reactions, particularly in the preparation of cupric salts and as a catalyst in coupling reactions. For instance, it is commonly utilized in the synthesis of aryl acetates through reactions with phenolic compounds. The ability of cupric acetate to facilitate the formation of carbon-carbon bonds has made it an essential tool in synthetic organic chemistry. In addition to its role in organic synthesis, cupric acetate monohydrate is significant in the field of electrochemistry. It has been employed as an electrolyte in electroplating processes, where it aids in the deposition of copper onto various substrates. The use of cupric acetate in this capacity is particularly advantageous due to its ability to maintain a stable solution, which is crucial for achieving uniform plating results. This property has made it a valuable component in the manufacturing of electronic components and decorative finishes. Cupric acetate monohydrate also finds applications in the textile industry, where it is used as a mordant in dyeing processes. Its ability to form stable complexes with dye molecules enhances the colorfastness and vibrancy of textiles. This characteristic has made it an essential agent in producing high-quality dyed fabrics. Moreover, the compound has been explored for its potential use in catalyzing oxidative processes in the dyeing industry. In recent years, cupric acetate monohydrate has gained attention in the field of materials science. Researchers have investigated its potential use in the synthesis of nanomaterials and nanoparticles, exploiting its properties to create novel materials with unique functionalities. The incorporation of cupric acetate in these processes has opened new avenues for developing advanced materials with applications in electronics, catalysis, and environmental remediation. Despite its beneficial applications, safety considerations are essential when handling cupric acetate monohydrate. The compound can pose health risks if ingested or inhaled, necessitating the implementation of appropriate safety measures during its use in industrial and laboratory settings. Ensuring proper ventilation, protective equipment, and adherence to safety guidelines is crucial for mitigating potential hazards. In summary, cupric acetate monohydrate is a versatile compound with significant implications in organic synthesis, electrochemistry, textiles, and materials science. Its discovery and subsequent applications have underscored the importance of copper compounds in various industrial processes, highlighting their role in advancing both scientific research and practical applications. References 2018. The Chemical and Products Database, a resource for exposure-relevant data on chemicals in consumer products. Scientific Data, 5. DOI: 10.1038/sdata.2018.125 1985. Acute oral toxicity and repellency of 933 chemicals to house and deer mice. Archives of Environmental Contamination and Toxicology, 14(1). URL: https://www.aphis.usda.gov/ws/nwrc/chem-effects-db/S_schafer851.pdf 1982. Journal of Animal Science. Journal of Animal Science, 55(337). URL: https://pubmed.ncbi.nlm.nih.gov/7142050 |
| Market Analysis Reports |