Online Database of Chemicals from Around the World
| Epochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.epochem.com | |||
 | +86 (21) 6760-1595 6760-1597 | |||
 | +86 (21) 6760-1605 | |||
 | seth_wang@epochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Yizheng East Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chinaeastchem.com | |||
 | +86 (514) 8796-1069 | |||
 | +86 (514) 8796-1106 | |||
 | fzj@chinaeastchem.com fang_yizhengeast@hotmail.com | |||
| Chemical manufacturer since 1989 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Changzhou Jintan ChengEn Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chengenchem.com | |||
 | +86 (519) 8019-0155 +86 13961176135 +86 13775159780 | |||
 | +86 (519) 8019-0155 | |||
 | sales@chengenchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Nanjing Hike Biological Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.njhkchem.com | |||
 | +86 (25) 5211-0956 | |||
 | +86 (25) 5211-0956 | |||
 | sophia@njhkchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: 17372769668 | |||
 | WhatsApp:17372769668 | |||
| Chemical distributor since 2021 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Kingchem Inc. | USA | |||
|---|---|---|---|---|
 | www.kingchem.com | |||
 | +1 (800) 211-4330 (201) 825-9988 | |||
 | +1 (201) 825-9148 | |||
 | customer-service@kingchem.com | |||
| Chemical manufacturer since 1994 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Aaron Chemistry GmbH | Germany | |||
|---|---|---|---|---|
 | www.aaron-chemistry.de | |||
 | +49 (8823) 917-521 | |||
 | +49 (8823) 917-523 | |||
 | sales@aaron-chemistry.de | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Nitro compound |
|---|---|
| Name | 2-Nitrobenzyl alcohol |
| Synonyms | o-Nitrobenzyl alcohol |
| Molecular Structure | 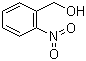 |
| Molecular Formula | C7H7NO3 |
| Molecular Weight | 153.14 |
| CAS Registry Number | 612-25-9 |
| EC Number | 210-302-6 |
| SMILES | C1=CC=C(C(=C1)CO)[N+](=O)[O-] |
| Melting point | 69-73 °C |
|---|---|
| Boiling point | 270 °C |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
2-Nitrobenzyl alcohol, an organic compound with the formula C7H7NO3, is part of the nitrobenzyl alcohol family. It was first synthesized in the early 20th century during studies on the nitration of aromatic compounds. The compound is produced by the reduction of 2-nitrobenzaldehyde or through the nitration of benzyl alcohol under controlled conditions. In organic synthesis, 2-nitrobenzyl alcohol serves as a key intermediate. It is used to synthesize various nitroaromatic compounds and can undergo transformations such as reduction, oxidation, and substitution. The nitro group can be reduced to an amino group, producing 2-aminobenzyl alcohol, which is a precursor for dyes, agrochemicals, and pharmaceuticals. Additionally, the hydroxyl group can be esterified or etherified to produce a variety of functional derivatives. 2-Nitrobenzyl alcohol is used in the pharmaceutical industry to synthesize active pharmaceutical ingredients (APIs) and intermediates. Its derivatives are explored for their potential therapeutic properties, including antimicrobial, antiviral, and anticancer activities. One of the notable applications of 2-nitrobenzyl alcohol is in photochemistry. The compound is used as a photolabile protecting group (PPG) for various functional groups in organic molecules. Upon exposure to ultraviolet (UV) light, 2-nitrobenzyl alcohol undergoes a photochemical reaction that releases the protected functional group. This property is utilized in the development of photoresponsive materials and in the controlled release of bioactive compounds in biochemical research. In materials science, 2-nitrobenzyl alcohol is employed in the development of advanced materials, including photoresists and photopolymers. Its photolabile properties make it useful in the fabrication of microelectronic devices and in photolithography processes. The compound is also explored for its potential in creating smart materials that respond to light stimuli. 2-Nitrobenzyl alcohol is widely used in academic and industrial research to study the mechanisms of photochemical and organic reactions. Researchers use it as a model compound to investigate the effects of nitro and hydroxyl groups on the reactivity and stability of aromatic compounds. In the agricultural sector, derivatives of 2-nitrobenzyl alcohol are investigated for their potential use as agrochemicals. Compounds derived from 2-nitrobenzyl alcohol can act as herbicides, insecticides, or fungicides, helping to protect crops from pests and diseases. References 2020. Sulfur-Mediated Decarboxylative Coupling of 2-Nitrobenzyl Alcohols and Arylacetic Acids. *Synlett*, 31(10). DOI: 10.1055/s-0040-1707113 2022. Transition-Metal-Free Synthesis of Quinolines from 2-Nitrobenzyl Alcohol in Water. *Science of Synthesis*. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-115-01180 2022. Niobium Oxide Supported on Cubic Spinel Cobalt Oxide as an Efficient Heterogeneous Catalyst for the Synthesis of Imines via Dehydrogenative Coupling of Amines and Alcohols. *Catalysis Letters*, 152(12). DOI: 10.1007/s10562-022-03943-2 |
| Market Analysis Reports |