Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Hangzhou Read Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hzread.com | |||
 | +86 (571) 8723-4293 8991-2636 | |||
 | +86 (571) 8723-4255 | |||
 | sales@hzread.com info@hzread.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Cangzhou Enke Pharma-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.enkepharma.com | |||
 | +86 (317) 510-5699 510-6597 +86 15533709196 | |||
 | +86 (317) 510-6596 | |||
 | sale@enkepharma.com enkepharma@126.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: ymzhao | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Nanjing Bic Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bicbiotech.com | |||
 | +86 18851183712 | |||
 | sales@bicbiotech.com | |||
 | QQ Chat | |||
 | WeChat: +8618851183712 | |||
 | WhatsApp:+8618851183712 | |||
| Chemical manufacturer since 2023 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Sinbond Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinbondpharm.com | |||
 | +86 (531) 8703-8285 +86 13583181986 +44 (208) 242-5518 | |||
 | +86 (531) 8703-8285 | |||
 | cici1124@gmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives >> Carboxylic esters and their derivatives |
|---|---|
| Name | Dimethyl (3-oxo-1,3-dihydroisobenzofuran-1-yl)phosphonate |
| Synonyms | (3-Oxo-1,3-dihydroisobenzofuran-1-yl)phosphonic acid dimethyl ester |
| Molecular Structure | 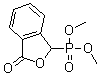 |
| Molecular Formula | C10H11O5P |
| Molecular Weight | 242.17 |
| CAS Registry Number | 61260-15-9 |
| EC Number | 964-929-1 |
| SMILES | COP(=O)(C1C2=CC=CC=C2C(=O)O1)OC |
| Density | 1.4$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 97-99 $degree$C (Expl.) |
| Boiling point | 402.0$+/-$45.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 210.4$+/-$49.1 $degree$C (Calc.)* |
| Index of refraction | 1.53 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Dimethyl (3-oxo-1,3-dihydroisobenzofuran-1-yl)phosphonate is a functionalized organophosphorus compound in which a phosphonate group is attached to a 1,3-dihydroisobenzofuran ring bearing a keto group at the 3-position. Compounds of this type are significant in organic synthesis due to the dual reactivity of the phosphonate moiety and the lactone ring system, enabling applications in carbon–carbon bond-forming reactions, heterocyclic synthesis, and the preparation of bioactive molecules. The phosphonate group (–PO(OCH3)2) acts as a nucleophilic site in reactions such as the Horner–Wadsworth–Emmons (HWE) olefination, while the 3-oxo-lactone provides electrophilic character and ring strain that can be exploited for nucleophilic additions or ring-opening transformations. Structurally, the molecule consists of a five-membered lactone (1,3-dihydroisobenzofuran) with a carbonyl group at the 3-position and a phosphonate ester attached at the 1-position. The lactone ring imparts rigidity and planarity, and the adjacent carbonyl group enhances electrophilicity at the 3-position. The phosphonate ester contains two methoxy groups attached to phosphorus, providing electron-withdrawing characteristics that stabilize the phosphorus center while making the α-carbon nucleophilic in HWE or Michael-type reactions. This combination of electrophilic and nucleophilic centers renders the compound a versatile intermediate in organic synthesis. The synthesis of dimethyl (3-oxo-1,3-dihydroisobenzofuran-1-yl)phosphonate is generally accomplished through the reaction of 3-hydroxyisobenzofuran-1-one derivatives with dialkyl phosphites under acidic or basic catalysis, often via a Michaelis–Arbuzov-type rearrangement or direct esterification. The reaction conditions are chosen to selectively introduce the dimethyl phosphonate group without affecting the lactone ring or the keto functionality. High-purity products can be obtained for subsequent synthetic transformations. In organic and medicinal chemistry, this compound is valuable as a precursor for the synthesis of α,β-unsaturated esters, phosphonate-containing heterocycles, and bioactive molecules. The phosphonate group is commonly used in Horner–Wadsworth–Emmons reactions to construct carbon–carbon double bonds with defined stereochemistry. The lactone ring can undergo nucleophilic ring-opening or condensation reactions to generate structurally diverse scaffolds for enzyme inhibitors, receptor ligands, or functional materials. The combination of phosphorus chemistry with a rigid bicyclic system allows chemists to access compounds with controlled stereochemistry and electronic properties. Beyond synthetic applications, dimethyl (3-oxo-1,3-dihydroisobenzofuran-1-yl)phosphonate serves as a useful intermediate for constructing phosphonate-containing heterocycles and bioactive derivatives. The α-carbon adjacent to the phosphorus atom is reactive toward electrophiles, enabling the formation of carbon–carbon bonds and further elaboration of the molecular framework. The lactone and keto functionalities can participate in ring expansions, condensations, or nucleophilic substitutions, providing multiple pathways for structural diversification. Overall, dimethyl (3-oxo-1,3-dihydroisobenzofuran-1-yl)phosphonate is a multifunctional organophosphorus intermediate combining a reactive phosphonate ester with a 3-oxo-lactone ring. Its electrophilic and nucleophilic centers, structural rigidity, and functional versatility make it a valuable building block in organic synthesis, heterocyclic chemistry, and the development of bioactive molecules. References 2025. Development and evaluation of a 99mTc-labeled olaparib analog for PARP imaging. EJNMMI Radiopharmacy and Chemistry. DOI: 10.1186/s41181-025-00373-4 2020. Synthetic Approaches to Contemporary Drugs that Contain the Cyclopropyl Moiety. Synthesis. DOI: 10.1055/s-0039-1690058 2007. Use of Stabilized Phthalide Anion Annulation Reactions in Synthesis: An Update. Synthesis. DOI: 10.1055/s-2007-965915 |
| Market Analysis Reports |