Online Database of Chemicals from Around the World
| Zhejiang Arts & Crafts Imp & Exp.co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zjartspharm.com | |||
 | +86 571 87238903 | |||
 | jocelynpan@zjarts.com | |||
| Chemical distributor since 1992 | ||||
| chemBlink Standard supplier since 2024 | ||||
| FCH Group | Ukraine | |||
|---|---|---|---|---|
 | www.fchgroup.net | |||
 | +38 (044) 228-9649 | |||
 | +38 (044) 495-8816 | |||
 | info@fchgroup.net | |||
| Chemical manufacturer | ||||
| IOLITEC Ionic Liquids Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.iolitec-usa.com | |||
 | +1 (205) 348-2831 | |||
 | +1 (205) 348-3510 | |||
 | info@iolitec-usa.com | |||
| Chemical manufacturer since 2002 | ||||
| Ionic Liquids Technologies GmbH & Co. KG | Germany | |||
|---|---|---|---|---|
 | www.iolitec.de | |||
 | +49 (7666) 913-929 | |||
 | +49 (7666) 912-9345 | |||
 | info@iolitec.de | |||
| Chemical manufacturer | ||||
| abcr GmbH | Germany | |||
|---|---|---|---|---|
 | www.abcr.de | |||
 | +49 (721) 950-610 | |||
 | +49 (721) 950-6180 | |||
 | info@abcr.de | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Imidazoles |
|---|---|
| Name | 1-(2-Hydroxyethyl)-3-methylimidazolium chloride |
| Synonyms | 3-(2-Hydroxyethyl)-1-methyl-1H-imidazolium chloride |
| Molecular Structure | 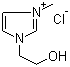 |
| Molecular Formula | C6H11N2O.Cl |
| Molecular Weight | 162.62 |
| CAS Registry Number | 61755-34-8 |
| EC Number | 695-725-8 |
| SMILES | C[N+]1=CN(C=C1)CCO.[Cl-] |
| Melting point | 86 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H319 Details | ||||||||||||||||
| Safety Statements | P264-P264+P265-P270-P280-P301+P317-P305+P351+P338-P330-P337+P317-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
1-(2-Hydroxyethyl)-3-methylimidazolium chloride is an important ionic liquid in the field of green chemistry. Its unique properties make it a versatile compound suitable for a variety of industrial and scientific applications. The discovery of ionic liquids, including 1-(2-hydroxyethyl)-3-methylimidazolium chloride, marked a breakthrough in the field of chemistry. Ionic liquids are salts that are liquid at room temperature and are known for their low volatility and high thermal stability. The specific compound, 1-(2-hydroxyethyl)-3-methylimidazolium chloride, often abbreviated as [HEMIM]Cl, was synthesized in the early 21st century as part of the ongoing research into environmentally friendly solvents. The molecular formula of 1-(2-hydroxyethyl)-3-methylimidazolium chloride is C6H11ClN2O. It consists of a chloride anion and a cation, with a hydroxyethyl group attached to the imidazole ring. This structure gives the compound several favorable properties: the hydroxyethyl group enhances its solubility in water; minimal vapor pressure reduces environmental pollution; suitability for high-temperature processes; and non-flammability, making it safer to handle than traditional solvents. 1-(2-Hydroxyethyl)-3-methylimidazolium chloride is widely used as a green solvent. Its ability to dissolve a wide range of substances makes it well suited for chemical synthesis, catalysis, and extraction processes. It offers an environmentally friendly alternative to volatile organic compounds (VOCs), reducing the ecological footprint of chemical manufacturing. This ionic liquid is also used in electrochemical devices. Its ionic conductivity and stability make it suitable for use in batteries, fuel cells, and supercapacitors. Researchers are exploring other potentials to increase the efficiency and lifetime of these devices, contributing to advances in energy storage technology. In the field of biomass processing, 1-(2-Hydroxyethyl)-3-methylimidazolium chloride is used to break down lignocellulosic materials. Its ability to dissolve cellulose and other biomass components facilitates the production of biofuels and biochemicals. This application is critical to developing sustainable energy and reducing dependence on fossil fuels. The biocompatibility of 1-(2-Hydroxyethyl)-3-methylimidazolium chloride has led to its widespread use in pharmaceuticals and biotechnology. It can be used as a mediator in drug delivery systems and enzyme-catalyzed reactions, improving the efficacy and stability of drug formulations. Its role in these areas is expanding as researchers discover new ways to exploit its properties. The compound is also being used in environmental remediation. Its ability to extract and recover heavy metals and other pollutants from water and soil makes it a promising agent for cleaning polluted environments. This application is in line with global efforts to address pollution and promote environmental sustainability. As a green solvent, 1-(2-hydroxyethyl)-3-methylimidazolium chloride minimizes the release of harmful VOCs, helping to purify the air and reduce environmental impact. Its ability to play a role in a variety of applications, from chemical synthesis to energy storage and environmental cleanup, highlights its versatility and usefulness in multiple industries. Non-flammable and stable, this ionic liquid improves safety in industrial and laboratory settings, reducing the risk of accidents and exposure to hazardous substances. References 2021. Cyclic carbonates synthesis by cycloaddition reaction of CO2 with epoxides in the presence of zinc-containing and ionic liquid catalysts. Journal of the Iranian Chemical Society, 18(12). DOI: 10.1007/s13738-021-02330-9 2013. Fundamentals of Ionic Liquids. Production of Biofuels and Chemicals with Ionic Liquids. DOI: 10.1007/978-94-007-7711-8_1 2007. Fabrication of silver nanoparticles via self-regulated reduction by 1-(2-hydroxyethyl)-3-methylimidazolium tetrafluoroborate. Korean Journal of Chemical Engineering, 24(6). DOI: 10.1007/s11814-007-0054-2 |
| Market Analysis Reports |