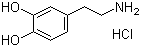
3-Hydroxytyramine hydrochloride, commonly referred to as dopamine hydrochloride, is the hydrochloride salt form of dopamine, a naturally occurring catecholamine and one of the principal neurotransmitters in both the central and peripheral nervous systems. Its structure includes a catechol ring (a benzene ring with two hydroxyl groups in adjacent positions) and an ethylamine side chain, making it a primary amine and a phenol derivative. The hydrochloride salt enhances its stability and solubility in aqueous solutions, facilitating its use in clinical settings.
Dopamine was first identified as a neurotransmitter in the 1950s, following earlier biochemical discoveries that had recognized it as a precursor to norepinephrine and epinephrine in the catecholamine biosynthetic pathway. Its physiological role extends across various regions of the brain, including the substantia nigra, where it plays a crucial part in motor control, as well as in the mesolimbic pathway, which is associated with reward and motivation. The formulation as a hydrochloride salt allows for medical administration, particularly in situations requiring intravenous delivery.
Clinically, dopamine hydrochloride is widely used as a cardiovascular drug, especially in emergency medicine and critical care. It is commonly employed in the treatment of shock, particularly in cases of myocardial infarction, trauma, sepsis, and heart failure, where it acts by stimulating dopaminergic and adrenergic receptors depending on the dosage administered. At low doses, it primarily stimulates dopaminergic receptors in the kidneys, resulting in vasodilation and increased renal perfusion. At moderate doses, it activates beta-1 adrenergic receptors, leading to enhanced cardiac output by increasing heart rate and contractility. At higher doses, alpha-adrenergic effects predominate, causing vasoconstriction and elevated blood pressure.
The compound is typically administered as an infusion under controlled conditions in hospital environments. Due to its rapid metabolism by monoamine oxidase (MAO) and catechol-O-methyltransferase (COMT), dopamine hydrochloride must be administered intravenously for systemic effects, as oral administration results in significant degradation before reaching the bloodstream. Its onset of action is within minutes, and the effects dissipate quickly after discontinuation, making it suitable for titration in acute care settings.
Beyond its application in critical care, 3-hydroxytyramine hydrochloride is also relevant in pharmacological research and neuroscience. It is used in studies investigating Parkinson’s disease, schizophrenia, and drug addiction, where dopamine dysregulation is implicated. The compound serves as a standard in analytical chemistry for the quantification of catecholamines in biological fluids via high-performance liquid chromatography (HPLC) and other electrochemical detection methods.
In chemical synthesis and research, dopamine hydrochloride has been employed in the preparation of functionalized surfaces, particularly through self-polymerization under alkaline conditions to form polydopamine coatings. This unique property has led to interest in biomedical engineering, nanotechnology, and material sciences, where polydopamine is used to modify surfaces for improved biocompatibility, adhesion, or as a scaffold for further chemical modification.
In storage and handling, dopamine hydrochloride must be kept in light-resistant containers and under controlled temperatures to avoid degradation. Exposure to light, air, or alkaline conditions can lead to oxidation, forming melanin-like pigments and reducing its pharmacological activity.
As a compound of major biological significance and clinical utility, 3-hydroxytyramine hydrochloride remains integral to both medicine and biochemical research. Its dual role as a neurotransmitter and a therapeutic agent underscores its importance in understanding and managing disorders of the nervous and cardiovascular systems.
References
2018. Dopamine promotes cathepsin B-mediated amyloid precursor protein degradation by reactive oxygen species-sensitive mechanism in neuronal cell. Molecular and Cellular Biochemistry, 451(1-2).
DOI: 10.1007/s11010-018-3460-3
2018. Dopamine D2 receptor-mediated circuit from the central amygdala to the bed nucleus of the stria terminalis regulates impulsive behavior. Proceedings of the National Academy of Sciences of the United States of America, 115(45).
DOI: 10.1073/pnas.1811664115
1976. Plasma dopamine-β-hydroxylase activity and the pressor effect of dopamine infusion in man. European Journal of Clinical Pharmacology, 10(2).
DOI: 10.1007/bf00558329
|

































































 GHS07;GHS09 Warning Details
GHS07;GHS09 Warning Details