Online Database of Chemicals from Around the World
| Discovery Fine Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.discofinechem.com | |||
 | +44 (1202) 874-517 | |||
 | +44 (845) 094-4385 | |||
 | pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Tianjin Zhongxin Chem-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tjzxchem.com | |||
 | +86 (22) 6688-0623 | |||
 | +86 (22) 8988-0739 ex 8030 | |||
 | sales@tjzxchem.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Anhui Royal Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.royal-chem.com | |||
 | +86 (25) 8663-9253 | |||
 | +86 (25) 5227-9193 | |||
 | zoey.zhao@royal-chem.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Georganics Ltd. | Slovakia | |||
|---|---|---|---|---|
 | www.georganics.sk | |||
 | +421 (33) 640-3132 | |||
 | +421 (33) 640-3133 | |||
 | georganics@georganics.sk | |||
| Chemical manufacturer since 1998 | ||||
| Classification | Organic raw materials >> Amino compound >> Cycloalkylamines, aromatic monoamines, aromatic polyamines and derivatives and salts |
|---|---|
| Name | 4-Methyl-N-methylaniline |
| Synonyms | N-Methyl-p-toluidine |
| Molecular Structure | 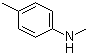 |
| Molecular Formula | C8H11N |
| Molecular Weight | 121.18 |
| CAS Registry Number | 623-08-5 |
| EC Number | 210-769-6 |
| SMILES | CC1=CC=C(C=C1)NC |
| Density | 0.958 |
|---|---|
| Boiling point | 212 °C |
| Refractive index | 1.5558-1.5578 |
| Flash point | 85 °C |
| Hazard Symbols |   GHS06;GHS08 Danger Details GHS06;GHS08 Danger Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H311-H331-H412 Details | ||||||||||||||||||||||||||||
| Safety Statements | P260-P261-P262-P264-P270-P271-P273-P280-P301+P316-P302+P352-P304+P340-P316-P319-P321-P330-P361+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| Transport Information | UN 2810 | ||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
4-Methyl-N-methylaniline, also known as N,N-dimethyl-p-toluidine, is an organic chemical compound with the formula C9H13N. It is a derivative of aniline, where the amine group is substituted with two methyl groups and a methyl group is attached to the aromatic ring. This compound is widely used as a curing agent and accelerator in polymer chemistry, especially in the production of dental materials and adhesives. The discovery of 4-Methyl-N-methylaniline is tied to advancements in organic chemistry in the early 20th century when researchers were exploring modifications to aniline and its derivatives for various industrial purposes. The addition of methyl groups to the nitrogen atom of aniline provided improved stability and solubility, making the compound more versatile for industrial applications. One of the main applications of 4-Methyl-N-methylaniline is in the polymerization of resins, particularly for dental restorations. In this role, it acts as a polymerization accelerator, helping to speed up the curing process of dental resins and adhesives. This is crucial in dental work, where the rapid hardening of materials like polymethyl methacrylate (PMMA) is necessary to complete procedures efficiently and ensure long-lasting results. In addition to dental applications, 4-Methyl-N-methylaniline is used in the formulation of certain adhesives, sealants, and coatings. Its ability to promote quick curing makes it useful in the automotive and construction industries, where fast-setting materials are required. The compound is also found in light-curing systems used for bonding and in the production of specialty polymers that require precise control of polymerization times. However, the use of 4-Methyl-N-methylaniline does raise concerns about toxicity. It is known to be a skin irritant and can cause allergic reactions in some individuals. Prolonged exposure may lead to more severe health effects, including damage to the liver and nervous system. Due to these risks, its use is regulated, and protective measures must be taken when handling it in industrial settings. The compound’s role in modern polymer chemistry, particularly in dental and adhesive applications, underscores its importance, but with ongoing attention to safety and environmental concerns. Research into alternatives with lower toxicity is ongoing as industries seek safer materials for widespread use. References 2024. Highly Effective Zirconia/γ-Alumina for Continuous Vapor Phase N-methylation of Aniline. Catalysis Letters. DOI: 10.1007/s10562-023-04568-9 2022. Photoredox-Mediated Desulfonylative Radical Reactions: An Excellent Approach Towards C-C and C-Heteroatom Bond Formation. Synthesis, 54(24). DOI: 10.1055/a-1900-8895 2021. Dual-function enzyme catalysis for enantioselective carbon-nitrogen bond formation. Nature Chemistry, 13(12). DOI: 10.1038/s41557-021-00794-z |
| Market Analysis Reports |