3-Chloro-1-propanol is an organic compound with the molecular formula C3H7ClO. It belongs to the class of haloalcohols, featuring both a hydroxyl group (–OH) and a chlorine atom attached to a three-carbon linear chain. Structurally, the chlorine atom is located at the terminal carbon opposite the hydroxyl group, making it a primary alcohol with a primary alkyl chloride function.
The compound has been known since the early to mid-20th century as part of research into chlorinated alcohols and their reactivity. Its synthesis can be accomplished through several methods, one of the most common being the chlorination of 1,3-propanediol under controlled conditions. Alternatively, it can be prepared via the hydrolysis of epichlorohydrin or as an intermediate in other synthetic pathways involving chlorinated alkanes.
3-Chloro-1-propanol has attracted attention due to its dual reactivity. The hydroxyl group allows for typical alcohol reactions, including esterification and oxidation, while the chlorine atom can undergo nucleophilic substitution to introduce a variety of functional groups. This bifunctionality makes it a valuable intermediate in organic synthesis, particularly in the preparation of heterocycles, pharmaceuticals, and polymer precursors.
One of the significant industrial applications of 3-chloro-1-propanol is its use as an intermediate in the production of pharmaceuticals and fine chemicals. It can serve as a starting material or building block for compounds such as amino alcohols, which are key constituents in various bioactive molecules. Its ability to participate in ring-closing reactions or be converted to epoxy compounds broadens its utility in synthetic organic chemistry.
In biochemical research, 3-chloro-1-propanol has been studied as a model compound for understanding the toxicity and metabolism of haloalcohols. Its metabolic fate in mammals includes oxidation by alcohol dehydrogenase enzymes to form the corresponding aldehyde, 3-chloropropionaldehyde, and subsequently 3-chloropropionic acid. These metabolites can interfere with cellular metabolism, particularly in the liver, leading to toxicological effects. As such, 3-chloro-1-propanol is considered a compound with potential health risks upon prolonged or high-level exposure.
Regulatory authorities have placed limits on the permissible levels of 3-chloro-1-propanol and its structural analogs in food and water due to concerns over genotoxicity. In particular, some chlorinated propanols, including 3-monochloropropane-1,2-diol (3-MCPD), are known contaminants in processed foods. While 3-chloro-1-propanol itself is less studied in this regard, its structural similarity has prompted research into its occurrence and potential formation during food processing.
In polymer chemistry, 3-chloro-1-propanol can act as a chain extender or reactive modifier in the synthesis of functionalized polyesters and polyurethanes. Its terminal alcohol group allows for incorporation into polymer backbones, while the chlorinated end can be modified to introduce other polar or reactive functionalities.
In laboratory settings, 3-chloro-1-propanol is used in synthetic protocols that require selective transformations. It has been employed in the preparation of sulfonates, azides, and amines through nucleophilic substitution, making it a versatile intermediate for producing functionalized three-carbon fragments. Additionally, it is used in the study of intramolecular cyclization reactions due to the spacing of the hydroxyl and halide groups, which enables the formation of three- or five-membered rings under appropriate conditions.
Safety considerations are important when handling 3-chloro-1-propanol. It is classified as harmful if swallowed or inhaled and can cause irritation to the skin and eyes. Proper protective equipment and ventilation are recommended when working with the compound in laboratory or industrial environments. Disposal must be conducted in accordance with local environmental regulations due to its potential toxicity and environmental persistence.
In summary, 3-chloro-1-propanol is a well-characterized haloalcohol that serves as an important intermediate in chemical synthesis, pharmaceutical production, and polymer research. Its bifunctional nature allows it to participate in a wide range of chemical reactions, and its reactivity profile has been explored extensively in both academic and industrial contexts. Despite its utility, appropriate caution is required due to its toxicological properties.
References
1978. Chlorohydrins in protein hydrolysates. Zeitschrift für Lebensmittel-Untersuchung und -Forschung.
URL: https://pubmed.ncbi.nlm.nih.gov/716635
2024. Cyanobacterial Pigments: Pharmaceutical and Nutraceutical Applications. Pharmaceutical and Nutraceutical Potential of Cyanobacteria.
DOI: 10.1007/978-3-031-45523-0_2
2019. Reaction of Isoselenocyanates and Alcohols with Subsequent Alkylation. Science of Synthesis.
URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-118-00803
|


















































































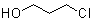
 GHS07 Warning Details
GHS07 Warning Details