Online Database of Chemicals from Around the World
| Beijing Eagle Sky Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.eagleskypharmatech.com | |||
 | +86 (10) 5979-9429 8875-5821 | |||
 | +86 (10) 5804-3698 | |||
 | sophia_818@126.com contact@eagleskypharmatech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Classification | Organic raw materials >> Ketone compound |
|---|---|
| Name | 2'-Hydroxy-4'-methoxypropiophenone |
| Synonyms | 1-(2-Hydroxy-4-methoxyphenyl)-1-propanone; NSC 35524 |
| Molecular Structure | 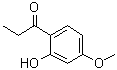 |
| Molecular Formula | C10H12O3 |
| Molecular Weight | 180.20 |
| CAS Registry Number | 6270-44-6 |
| EC Number | 621-243-4 |
| SMILES | CCC(=O)C1=C(C=C(C=C1)OC)O |
| Solubility | Very slightly soluble (0.85 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.126±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Melting point | 66 °C** |
| Index of Refraction | 1.531, Calc.* |
| Boiling Point | 314.7±22.0 °C (760 mmHg), Calc.* |
| Flash Point | 124.0±15.8 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2019 ACD/Labs) |
| ** | Gobbi, Silvia |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
2'-Hydroxy-4'-methoxypropiophenone is an organic compound that belongs to the class of substituted aromatic ketones. This compound contains a phenyl group substituted with a hydroxyl group at the 2'-position and a methoxy group at the 4'-position of the aromatic ring, along with a propiophenone moiety. The specific arrangement of functional groups makes 2'-hydroxy-4'-methoxypropiophenone a molecule of interest for its potential applications in various fields of organic synthesis and pharmaceutical chemistry. The discovery of 2'-hydroxy-4'-methoxypropiophenone is linked to efforts in designing new chemical entities with potential bioactive properties. The functional groups attached to the aromatic ring, namely the hydroxyl and methoxy substituents, can significantly influence the compound's electronic properties, such as its ability to participate in electrophilic substitution reactions or interact with biological targets. This structure suggests that 2'-hydroxy-4'-methoxypropiophenone might exhibit desirable pharmacological activity due to its ability to form hydrogen bonds and its potential for modulating the function of various biomolecules. In terms of applications, 2'-hydroxy-4'-methoxypropiophenone is often explored in the context of synthetic organic chemistry and drug discovery. The hydroxyl group can confer solubility and improve interactions with biological systems, while the methoxy group may enhance the compound's lipophilicity, making it suitable for potential drug design. Additionally, the propiophenone structure, which is a common building block in the synthesis of biologically active compounds, allows for easy functionalization to yield derivatives with varied pharmacological profiles. One of the main applications of 2'-hydroxy-4'-methoxypropiophenone is in the development of compounds with anti-inflammatory, anticancer, and antimicrobial activities. Research has shown that similar compounds can interact with enzymes, receptors, and other proteins involved in cellular signaling pathways, potentially leading to therapeutic effects. Furthermore, 2'-hydroxy-4'-methoxypropiophenone may serve as a precursor for the synthesis of more complex molecules with enhanced potency and selectivity. In conclusion, 2'-hydroxy-4'-methoxypropiophenone represents a promising scaffold in organic synthesis and drug development. Its unique structural features offer potential for various chemical modifications, making it a valuable tool for the creation of new drug candidates with improved biological activity. References 1975. Isolation of 2′-Hydroxy-4′-methoxy propiophenone fromTribolium castaneumandT confusum. Agricultural and Biological Chemistry. DOI: 10.1080/00021369.1975.10861843 1986. Prostaglandin synthetase inhibitors in insect defensive secretions. The Science of Nature. DOI: 10.1007/bf00399245 1987. Defensive chemistry of the flour beetleTribolium brevicornis (LeC): : Presence of known and potential prostaglandin synthetase inhibitors. Journal of Chemical Ecology. DOI: 24302339 |
| Market Analysis Reports |