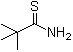
2,2-Dimethylpropanethioamide, also known as N,N-diisopropylthioacetamide, was first synthesized through organic chemistry research aimed at developing sulfur-containing compounds with potential industrial and pharmaceutical applications. Chemists focused on modifying the structure of propanethioamide—a thioamide compound— to introduce two methyl groups at the alpha position while preserving the thioamide functional group. Through synthetic routes involving the alkylation of suitable thioamide precursors, 2,2-Dimethylpropanethioamide was successfully produced. Its discovery marked a significant advancement in organic synthesis, providing a versatile building block for the synthesis of various organic compounds with applications in diverse fields.
2,2-Dimethylpropanethioamide and its derivatives serve as key intermediates in the synthesis of pharmaceutical compounds, including thioamides, sulfonamides, and heterocyclic molecules. These compounds exhibit diverse pharmacological activities, such as antimicrobial, antiviral, and anticancer properties, making them promising candidates for drug development. Some derivatives of 2,2-Dimethylpropanethioamide are utilized as chiral auxiliaries in asymmetric synthesis to control the stereochemistry of reactions and enhance the selectivity of chemical transformations. They facilitate the synthesis of chiral pharmaceuticals with improved efficacy and reduced side effects.
2,2-Dimethylpropanethioamide derivatives are employed in organic synthesis as sulfur-containing building blocks for the construction of complex molecules. They participate in various chemical transformations, such as nucleophilic substitution, oxidation, and reduction, enabling the synthesis of diverse organic compounds with specific functionalities. Some derivatives of 2,2-Dimethylpropanethioamide serve as ligands in transition metal catalysis, facilitating chemical reactions through coordination with metal centers. These catalyst systems exhibit high efficiency and selectivity in a wide range of transformations, including cross-coupling reactions, asymmetric hydrogenation, and C-H activation.
2,2-Dimethylpropanethioamide derivatives are utilized as additives in polymer synthesis to modify the properties of polymers, such as thermal stability, flame retardancy, and mechanical strength. They act as chain extenders, crosslinking agents, or stabilizers, enhancing the performance of polymers in various applications, including plastics, coatings, and adhesives. Some derivatives of 2,2-Dimethylpropanethioamide are employed in surface modification techniques to functionalize materials, such as nanoparticles, membranes, and surfaces of biomedical implants. These compounds facilitate the attachment of functional groups or biomolecules, enabling tailored surface properties for specific applications in biotechnology, catalysis, and medical devices.
References
1988. Activation of α-sp3 centres toward electrophilic substitution in alcohols and amines. Proceedings of the Indian Academy of Sciences - Chemical Sciences, 100(2).
DOI: 10.1007/bf02839447
2014. Dabrafenib. Pharmaceutical Substances.
URL: KD-04-0183
|































 GHS07 Warning Details
GHS07 Warning Details