Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Nanjing MSN Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.msnchem.com | |||
 | +86 (25) 5701-5632 +86 18013328036 | |||
 | +86 (25) 5701-9235 | |||
 | info@msnchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Rintech, Inc. | USA | |||
|---|---|---|---|---|
 | www.rintechinc.com | |||
 | +1 (301) 987-1980 | |||
 | +1 (301) 987-1981 | |||
 | info@rintechinc.com | |||
| Chemical manufacturer since 1997 | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Frontier Scientific Services, Inc. | USA | |||
|---|---|---|---|---|
 | www.frontierssi.com | |||
 | +1 (302) 266-6891 (888) 577-2734 | |||
 | +1 (302) 266-8296 | |||
 | customerservice@frontierssi.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Organic raw materials >> Amino compound >> Cycloalkylamines, aromatic monoamines, aromatic polyamines and derivatives and salts |
|---|---|
| Name | 2-tert-Butylaniline |
| Molecular Structure | 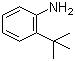 |
| Molecular Formula | C10H15N |
| Molecular Weight | 149.23 |
| CAS Registry Number | 6310-21-0 |
| EC Number | 228-634-5 |
| SMILES | CC(C)(C)C1=CC=CC=C1N |
| Density | 0.9±0.1 g/cm3, Calc.*, 0.957 g/mL (Expl.) |
|---|---|
| Melting point | -60 °C (Expl.) |
| Index of Refraction | 1.529, Calc.*, 1.545 (Expl.) |
| Boiling Point | 234.6±9.0 °C (760 mmHg), Calc.*, 263.9-265.2 °C (Expl.) |
| Flash Point | 102.2 °C, Calc.*, 102 °C (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P305+P351+P338 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
2-tert-Butylaniline, with the chemical formula C10H15N, is an aromatic amine compound that consists of a benzene ring with a tert-butyl group and an amino group (-NH2) attached to it at the 2-position. It is one of the isomers of butylaniline, differing from other isomers in the position of the substituents on the benzene ring. The discovery and synthesis of 2-tert-butylaniline are part of the broader development of substituted aniline derivatives in the late 19th and early 20th centuries. Aniline and its derivatives have been of significant interest due to their role as key intermediates in the chemical and pharmaceutical industries. Over the years, the synthesis of various substituted aniline derivatives, including 2-tert-butylaniline, has been explored for their industrial applications. 2-tert-Butylaniline is primarily used in the chemical industry as a starting material for the synthesis of various organic compounds. One of its major uses is in the production of dyes and pigments, where it serves as an intermediate in the synthesis of azo dyes. Azo dyes are widely used in the textile industry for coloring fabrics and are also applied in other industries such as food and cosmetics. In addition to its use in dye manufacturing, 2-tert-butylaniline is employed in the synthesis of certain pharmaceuticals and agrochemicals. The presence of both the amino group and the bulky tert-butyl group makes it a useful intermediate in the creation of compounds with specific steric and electronic properties. These properties can be useful in the development of more targeted drugs and chemical products. 2-tert-butylaniline is also a valuable reagent in research, particularly in organic synthesis. The amino group can participate in a variety of chemical reactions, such as nucleophilic substitution and condensation reactions, to form more complex molecules. These reactions are essential in the development of new materials and in the study of chemical reactivity in aromatic systems. In conclusion, 2-tert-butylaniline is an important chemical intermediate with applications in the production of dyes, pharmaceuticals, and agrochemicals. Its utility in organic synthesis and its role as a precursor to various industrial compounds underscore its importance in both research and commercial applications. References 2011. Synthesis and characterization of highly soluble aromatic polyimides. Chinese Journal of Polymer Science, 29(4). DOI: 10.1007/s10118-011-1058-2 2016. Synthesis and characterization of polyimides from 4,4′-(3-(tert-butyl)-4-aminophenoxy)diphenyl ether. Journal of Polymer Research, 24(1). DOI: 10.1007/s10965-016-1168-1 2022. Trifarotene. Pharmaceutical Substances. URL: https://pharmaceutical-substances.thieme.com/ps/search-results?docUri=KD-20-0249 |
| Market Analysis Reports |