Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Henan Haohai Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.haohaichem.com | |||
 | +86 (371) 6391-6105 | |||
 | +86 (371) 6393-2448 | |||
 | 3h@haohaichem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| F&F Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fnfchem.com | |||
 | +86 (10) 6444-6910 | |||
 | +86 (10) 6444-6915 | |||
 | heyifnfchem@163.com heyi@fnfchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| HBCChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.hbcchem-inc.com | |||
 | +1 (510) 219-6317 | |||
 | +1 (510) 675-0318 | |||
 | sales@hbcchem-inc.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Lontr Science & Technology Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lontr.com | |||
 | +86 (25) 8434-1022 | |||
 | +86 (25) 8434-1022 | |||
 | zjb298680@163.com zjb29868@hotmail.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Advint Pharmaceutical Pvt Ltd. | India | |||
|---|---|---|---|---|
 | www.advintpharma.in | |||
 | 9773685656 | |||
 | sales@advintpharma.com | |||
| Chemical distributor since 2021 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Frontier Scientific Services, Inc. | USA | |||
|---|---|---|---|---|
 | www.frontierssi.com | |||
 | +1 (302) 266-6891 (888) 577-2734 | |||
 | +1 (302) 266-8296 | |||
 | customerservice@frontierssi.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Fatty aldehyde (containing acetal, hemiacetal) |
|---|---|
| Name | 2,3-Dichlorobenzaldehyde |
| Molecular Structure | 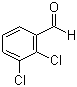 |
| Molecular Formula | C7H4Cl2O |
| Molecular Weight | 175.01 |
| CAS Registry Number | 6334-18-5 (31155-09-6) |
| EC Number | 228-711-3 |
| SMILES | C1=CC(=C(C(=C1)Cl)Cl)C=O |
| Density | 1.4±0.1 g/cm3, Calc.*, 1.33 g/mL (Expl.) |
|---|---|
| Melting point | 64-67 °C (Expl.) |
| Index of Refraction | 1.601, Calc.* |
| Boiling Point | 242.9±20.0 °C (760 mmHg), Calc.*, 143-145 °C (Expl.) |
| Flash Point | 99.2±22.3 °C, Calc.*, 135 °C (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H314-H315-H335-H412 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P261-P264-P271-P273-P280-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P354+P338-P316-P319-P321-P332+P317-P362+P364-P363-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 3261 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|
2,3-Dichlorobenzaldehyde, with the molecular formula C7H4Cl2O, is an aromatic compound consisting of a benzene ring substituted with two chlorine atoms at the 2- and 3-positions and an aldehyde group at the 1-position. This compound has garnered attention due to its versatility in organic synthesis and applications in various fields, including pharmaceuticals, agrochemicals, and materials science. The compound was first synthesized through chlorination processes applied to benzaldehyde derivatives. Early chlorination techniques involved the use of reagents such as chlorine gas or sulfuryl chloride under controlled conditions to selectively introduce chlorine atoms into aromatic rings. The development of such halogenated aldehydes provided chemists with intermediates for more complex synthetic pathways. 2,3-Dichlorobenzaldehyde is a valuable building block in the synthesis of bioactive molecules. In pharmaceutical research, it serves as a precursor for the development of antibacterial, antifungal, and anti-inflammatory agents. The dichloro substitution pattern enhances the reactivity and pharmacological properties of derivatives, making it a useful scaffold for medicinal chemistry programs. In the field of agrochemicals, 2,3-dichlorobenzaldehyde is employed in the synthesis of herbicides, pesticides, and fungicides. Its structural features contribute to the biological activity of these compounds, helping to inhibit the growth of unwanted pests and pathogens in agriculture. Additionally, 2,3-dichlorobenzaldehyde finds use in material science, particularly in the development of liquid crystals and polymers. The compound’s aromatic structure and electron-withdrawing chlorine atoms impart desirable properties such as rigidity and thermal stability to the resulting materials. Industrial production methods for 2,3-dichlorobenzaldehyde have evolved to prioritize efficiency and environmental safety. Modern techniques utilize catalytic processes and optimized reaction conditions to minimize by-products and waste. Advances in green chemistry have also explored more sustainable methods for halogenation reactions. In summary, 2,3-dichlorobenzaldehyde is a key intermediate in the synthesis of pharmaceuticals, agrochemicals, and advanced materials. Its discovery and applications highlight the importance of halogenated aromatic compounds in modern chemistry. References 2011. Intermolecular interactions and unexpected isostructurality in the crystal structures of the dichlorobenzaldehyde isomers. Acta crystallographica. Section B, Structural science, 67(5). DOI: 10.1107/s0108768111035786 2012. Synthesis of tetrazolodiazepines by a five-centered four-component azide Ugi reaction. Scope and limitations. Russian Chemical Bulletin, 61(8). DOI: 10.1007/s11172-012-0214-3 2016. Catalyst-Free Synthesis of 2-Arylbenzothiazoles in an Air/DMSO Oxidant System. Synlett, 27(7). DOI: 10.1055/s-0035-1561575 |
| Market Analysis Reports |