Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Ring Specialty Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.ringchemicals.com | |||
 | +1 (416) 493-6870 | |||
 | info@ringchemicals.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shuyang Huatai Chemical Factory | China | |||
|---|---|---|---|---|
 | www.huatai-chem.com | |||
 | +86 (527) 8331-3628 | |||
 | +86 (527) 8331-3638 | |||
 | wsq@huatai-chem.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Chemical pesticide >> Insecticide intermediate |
|---|---|
| Name | Methyl 3,3-dimethylpent-4-enoate |
| Synonyms | 3,3-Dimethyl-4-pentenoic acid methyl ester |
| Molecular Structure | 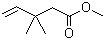 |
| Molecular Formula | C8H14O2 |
| Molecular Weight | 142.20 |
| CAS Registry Number | 63721-05-1 |
| EC Number | 264-431-8 |
| SMILES | CC(C)(CC(=O)OC)C=C |
| Density | 0.899 |
|---|---|
| Refractive index | 1.423 |
| Flash point | 107 °F |
| Hazard Symbols |  GHS02 Warning Details GHS02 Warning Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226 Details | ||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P280-P303+P361+P353-P370+P378-P403+P235-P501 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
| Transport Information | UN 3272 | ||||||||
| SDS | Available | ||||||||
|
Methyl 3,3-dimethylpent-4-enoate, an organic compound, was first synthesized during the exploration of esterification reactions involving unsaturated alcohols and carboxylic acids. The compound features a distinct structure with a methyl ester group and an alkenyl chain, making it valuable for various applications. Its synthesis was confirmed through techniques such as nuclear magnetic resonance (NMR) and mass spectrometry (MS), which verified its chemical composition and structure. The discovery and subsequent synthesis of methyl 3,3-dimethylpent-4-enoate have been significant in organic chemistry, particularly in the development of new materials and pharmaceuticals. Methyl 3,3-dimethylpent-4-enoate is often used as an intermediate in organic synthesis. Its unique structure allows for various chemical transformations, making it a useful building block for creating complex molecules. This compound is valuable in the synthesis of fine chemicals, agrochemicals, and other specialized organic compounds. In pharmaceutical research, methyl 3,3-dimethylpent-4-enoate serves as a precursor for synthesizing bioactive molecules. Its ability to undergo various chemical reactions enables the development of novel drug candidates. Researchers utilize this compound to create new therapeutic agents targeting a range of medical conditions, including inflammation, infections, and metabolic disorders. The compound's reactivity makes it suitable for use in polymer science. Methyl 3,3-dimethylpent-4-enoate can be incorporated into polymer backbones, leading to the creation of specialized materials with desired properties. These materials find applications in coatings, adhesives, and other high-performance products where specific chemical resistance or mechanical properties are required. Due to its ester functionality, methyl 3,3-dimethylpent-4-enoate is utilized in the flavor and fragrance industry. It contributes to the synthesis of various aroma compounds, adding fruity or floral notes to fragrances and flavorings. Its use enhances the sensory characteristics of consumer products such as perfumes, cosmetics, and food items. Methyl 3,3-dimethylpent-4-enoate is employed in the development of agrochemicals, including herbicides and insecticides. Its chemical properties facilitate the creation of active ingredients that protect crops from pests and diseases, thereby improving agricultural productivity and sustainability. References 2002. Novel convenient synthesis and chemoselective derivatization of 1-sila-4-stannacyclohexanes. Silicon Chemistry. DOI: 10.1023/b:silc.0000025575.46941.99 2012. Functional Group Tolerable Synthesis of Allylsilanes through Copper-Catalyzed γ-Selective Allyl-Alkyl Coupling between Allylic Phosphates and Alkylboranes. Synthesis. DOI: 10.1055/s-0031-1290818 2024. A study of alkene disulfonoxylation. Medicinal chemistry research : an international journal for rapid communications on design and mechanisms of action of biologically active agents, 33(5). DOI: 10.1007/s00044-024-03239-7 |
| Market Analysis Reports |