Online Database of Chemicals from Around the World
| Shanghai ZaiQi Bio-Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemzq.com | |||
 | +86 (21) 5482-4098 | |||
 | +86 (21) 5482-4069 | |||
 | sales1@chemzq.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Rhodia (shanghai) International Trading Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rhodia.com | |||
 | +86 (21) 2408-9317 +86 13601753458 | |||
 | +86 (21) 5483-0035 | |||
 | hank.xue@ap.rhodia.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Digital Specialty Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.digitalchem.com | |||
 | +1 (603) 563-5060 | |||
 | +1 (603) 563-9288 | |||
 | sales@digitalchem.com | |||
| Chemical manufacturer since 1987 | ||||
| Achemica | Switzerland | |||
|---|---|---|---|---|
 | www.achemica.com | |||
 | +41 (24) 466-2929 | |||
 | +41 (24) 466-2800 | |||
 | contact@achemica.com | |||
| Chemical manufacturer since 2010 | ||||
| Classification | Chemical reagent >> Organic reagent >> Phosphonate / phosphonate |
|---|---|
| Name | Dipentyl pentylphosphonate |
| Synonyms | Pentylphosphonic acid dipentyl ester |
| Molecular Structure | 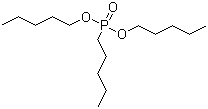 |
| Molecular Formula | C15H33O3P |
| Molecular Weight | 292.39 |
| CAS Registry Number | 6418-56-0 |
| EC Number | 229-144-4 |
| SMILES | CCCCCOP(=O)(CCCCC)OCCCCC |
| Density | 0.9±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 364.8±11.0 °C 760 mmHg (Calc.)* |
| Flash point | 188.1±39.6 °C (Calc.)* |
| Index of refraction | 1.436 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
2‑Dipentyl pentylphosphonate (abbreviated DP[PP] and also known as diamyl amylphosphonate, DAAP) is an organophosphorus compound used primarily as an extractant in nuclear‑analytical chemistry. Its molecular formula is C15H33O3P. The substance gained prominence through its incorporation into the UTEVA (Uranium and TEtraValent Actinides) extraction‑chromatography resin, which was developed to selectively separate uranium(VI) and tetravalent actinides (e.g., Th(IV), Np(IV), Pu(IV)). The extractant is impregnated onto an inert support, and its selectivity arises from its strong complexation of actinide nitrato species, particularly at elevated nitrate concentrations. As the nitrate concentration increases, the binding affinity of DP[PP] toward these actinides increases sharply, enabling efficient extraction under highly acidic conditions. In application, UTEVA resin containing DP[PP] has been used in both environmental and radiochemical workflows. One notable use is in the purification of scandium‑44 for positron emission tomography (PET): after proton irradiation of calcium targets, 44Sc(III) can be separated from large amounts of calcium using DP[PP]‑based resin under hydrochloric acid conditions. The method yields over 80 percent recovery within about 20 minutes and produces eluates with suitable chemical purity for radiolabeling. DP[PP] is also applied in analytical nuclear chemistry for actinide determination. The resin’s selectivity allows for the rapid separation of uranium and plutonium from complex matrices. For example, it has been used prior to thermal‑ionization mass spectrometry, significantly accelerating isolation times compared with older ion‑exchange techniques. Beyond separation of U and Pu, DP[PP] has been demonstrated in solvent‑extraction systems to isolate hexavalent americium (Am(VI)). When incorporated into an organic phase (e.g., in dodecane), 1 M DAAP can achieve high separation factors for Am(VI) over lanthanides under strongly acidic conditions. After extraction, Am(VI) may be selectively stripped by reducing it back to Am(III) using dilute acid solutions. From a safety and handling perspective, DP[PP] is used as a functional extractant and thus typically handled as part of a supported resin or dissolved in organic diluents. Appropriate laboratory precautions involve working in well-ventilated areas, using gloves and eye protection, and avoiding exposure to strong oxidants or extreme conditions that could degrade the phosphonate. DP[PP] has therefore become a cornerstone extractant in nuclear‑analytical chemistry, combining robustness, selectivity, and compatibility with widely used chromatographic formats. References Valdovinos HF, Hernandez R, Barnhart TE, Graves S, Cai W, Nickles RJ (2014) Separation of cyclotron‑produced 44Sc from a natural calcium target using a dipentyl pentylphosphonate functionalized extraction resin. Applied Radiation and Isotopes 95: 23–29 DOI: 10.1016/j.apradiso.2014.09.020 Goodall P, Lythgoe C (1999) Rapid separation of uranium and plutonium by extraction chromatography for determination by thermal ionisation mass spectrometry. Analyst 124: 263–269 DOI: 10.1039/A809219B Mincher BJ, Martin LR, Schmitt NC (2012) Diamylamylphosphonate Solvent Extraction of Am(VI) from Nuclear Fuel Raffinate Simulant Solution. Solvent Extraction and Ion Exchange 30(5): 445–456 DOI: 10.1080/07366299.2012.671108 |
| Market Analysis Reports |