Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Alfa Chemistry | USA | |||
|---|---|---|---|---|
 | www.alfa-chemistry.com | |||
 | +1 (201) 478-8534 | |||
 | +1 (516) 927-0118 | |||
 | inquiry@alfa-chemistry.com | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | API >> Antibiotics |
|---|---|
| Name | Pimeprofen |
| Synonyms | BE 100; Ibuprofen piconol; Staderm; U 75630; Vesicum |
| Molecular Structure | 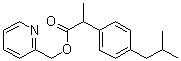 |
| Molecular Formula | C19H23NO2 |
| Molecular Weight | 297.39 |
| CAS Registry Number | 64622-45-3 (112017-99-9) |
| SMILES | CC(C)CC1=CC=C(C=C1)C(C)C(=O)OCC2=CC=CC=N2 |
| Solubility | Practically insoluble (0.039 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.063±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Boiling point | 410.7±30.0 °C 760 mmHg (Calc.)* |
| Flash point | 202.2±24.6 °C (Calc.)* |
| Index of refraction | 1.545 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
Pimeprofen is a nonsteroidal anti-inflammatory drug (NSAID) belonging to the propionic acid class, known for its analgesic, anti-inflammatory, and antipyretic properties. It was developed to provide effective relief from pain and inflammation associated with musculoskeletal conditions while aiming to minimize some of the adverse effects commonly observed with earlier NSAIDs. The molecular formula of pimeprofen is C15H14O3, and it is structurally characterized by a propionic acid backbone attached to an aromatic ring system, contributing to its pharmacological activity. The compound emerged in the pharmaceutical landscape in the 1970s during a period of extensive research on NSAIDs that could effectively inhibit the cyclooxygenase (COX) enzymes responsible for prostaglandin synthesis. Prostaglandins play a key role in mediating inflammation, pain, and fever, and the inhibition of COX enzymes has been the primary therapeutic target for NSAIDs. Pimeprofen specifically acts as a reversible inhibitor of both COX-1 and COX-2 isoforms, reducing the production of pro-inflammatory prostaglandins and thereby alleviating symptoms such as swelling, pain, and fever. Clinically, pimeprofen has been used in the treatment of inflammatory disorders including rheumatoid arthritis, osteoarthritis, and other conditions involving acute or chronic musculoskeletal pain. By mitigating inflammation and providing analgesia, it improves patient mobility and quality of life. Its antipyretic effect also allows it to be used in the management of fever in various inflammatory and infectious conditions. Pharmacokinetically, pimeprofen is typically administered orally in tablet form. After ingestion, it is absorbed through the gastrointestinal tract and undergoes hepatic metabolism, primarily via phase I oxidative pathways and phase II conjugation reactions. The drug’s metabolites are excreted through renal pathways. The elimination half-life of pimeprofen allows for dosing schedules generally ranging from two to three times daily to maintain therapeutic plasma concentrations. While effective, pimeprofen shares safety concerns common to NSAIDs. Gastrointestinal irritation is a significant adverse effect, potentially leading to ulceration or bleeding with prolonged use. Patients may also experience hypersensitivity reactions, renal impairment, and cardiovascular risks, especially when used at higher doses or in vulnerable populations. Thus, careful patient selection and monitoring are essential during pimeprofen therapy. In terms of chemical properties, pimeprofen is a crystalline solid at room temperature, sparingly soluble in water but soluble in organic solvents like ethanol and dimethyl sulfoxide. Its stability under physiological conditions contributes to its effective pharmacological profile. Analytical methods for pimeprofen include high-performance liquid chromatography (HPLC) for quantifying the drug in pharmaceutical formulations and biological fluids. Ultraviolet-visible (UV-Vis) spectrophotometry is used to assess purity and concentration, while mass spectrometry (MS) provides structural confirmation and metabolite identification. These analytical techniques ensure quality control during manufacturing and support pharmacokinetic and bioequivalence studies. Research into pimeprofen has also explored its potential antioxidant properties and effects on inflammatory mediators beyond COX inhibition, although these areas require further study to establish clinical relevance. Its comparison with other propionic acid NSAIDs such as ibuprofen and naproxen has demonstrated a similar efficacy profile, with some differences in tolerability and pharmacokinetics influencing therapeutic choices. In industrial and pharmaceutical settings, pimeprofen is manufactured through established organic synthesis routes involving the formation of the propionic acid core and subsequent aromatic substitution reactions. Quality standards ensure the purity, potency, and safety of the final product in compliance with regulatory guidelines. In summary, pimeprofen is a propionic acid-derived NSAID that exerts anti-inflammatory, analgesic, and antipyretic effects primarily through inhibition of COX enzymes and reduction of prostaglandin synthesis. Its clinical applications focus on managing musculoskeletal inflammatory conditions, with a pharmacokinetic profile suited for oral administration and routine dosing. Safety considerations typical of NSAIDs apply, necessitating careful use in susceptible patients. Analytical and manufacturing processes support its consistent availability and quality as a therapeutic agent. References 2023. A preclinical secondary pharmacology resource illuminates target-adverse drug reaction associations of marketed drugs. Nature Communications, 14(7). DOI: 10.1038/s41467-023-40064-9 1987. High-performance liquid chromatographic determination of pimeprofen and its metabolite ibuprofen in sheep plasma and lymph. Journal of Chromatography B: Biomedical Sciences and Applications, 413. DOI: 10.1016/0378-4347(87)80046-4 |
| Market Analysis Reports |