Online Database of Chemicals from Around the World
| BrightGene Bio-medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bright-gene.com | |||
 | +86 (512) 6255-1801 6255-1767 +86 13812696362 | |||
 | +86 (512) 6255-1799 | |||
 | kevinwan@bright-gene.com kevinwan0203@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Waterstone Pharmaceuticals, Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.waterstonechina.com | |||
 | +86 (27) 8753-1661 | |||
 | wangjiajun@waterstonepharma.com | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Shanghai Min-biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.min-biotech.com | |||
 | +86 15190045345 | |||
 | sales@min-biotech.com | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | API >> Antibiotics |
|---|---|
| Name | VcMMAE |
| Synonyms | Maleimidocaproyl-valine-citrulline-p-aminobenzoyloxycarbonyl-monomethyl auristatin E; Vedotin |
| Molecular Structure | 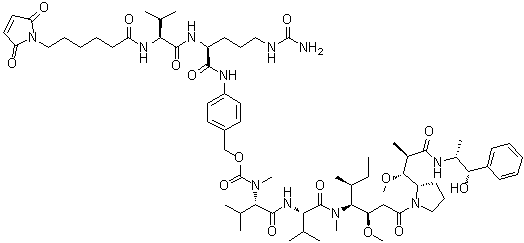 |
| Molecular Formula | C68H105N11O15 |
| Molecular Weight | 1316.63 |
| CAS Registry Number | 646502-53-6 |
| EC Number | 809-635-5 |
| SMILES | CC[C@H](C)[C@@H]([C@@H](CC(=O)N1CCC[C@H]1[C@@H]([C@@H](C)C(=O)N[C@H](C)[C@H](C2=CC=CC=C2)O)OC)OC)N(C)C(=O)[C@H](C(C)C)NC(=O)[C@H](C(C)C)N(C)C(=O)OCC3=CC=C(C=C3)NC(=O)[C@H](CCCNC(=O)N)NC(=O)[C@H](C(C)C)NC(=O)CCCCCN4C(=O)C=CC4=O |
| Density | 1.2±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.556, Calc.* |
| Boiling Point | 1347.6±65.0 °C (760 mmHg), Calc.* |
| Flash Point | 768.8±34.3 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS06;GHS07 Danger Details GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H300+H310+H330-H300-H310-H315-H319-H330-H335 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P261-P262-P264-P264+P265-P270-P271-P280-P284-P301+P316-P302+P352-P304+P340-P305+P351+P338-P316-P319-P320-P321-P330-P332+P317-P337+P317-P361+P364-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
VcMMAE, also known as monomethyl auristatin E, is a synthetic derivative of dolastatin 10, a natural product isolated from the marine mollusk Dolabella auricularia. It was discovered through extensive chemical modifications aimed at enhancing its cytotoxic properties and optimizing its therapeutic potential. VcMMAE was developed as a potent cytotoxic agent capable of selectively targeting cancer cells by inhibiting microtubule assembly and inducing cell cycle arrest and apoptosis. VcMMAE is widely used in the development of antibody-drug conjugates (ADCs), a class of targeted cancer therapies that combine the specificity of monoclonal antibodies with the cytotoxicity of small-molecule drugs. In ADCs, VcMMAE is conjugated to monoclonal antibodies that selectively bind to tumor-specific antigens expressed on cancer cells. Upon internalization, VcMMAE is released from the antibody and exerts its cytotoxic effects, leading to the selective destruction of cancer cells while sparing healthy tissues. ADCs containing VcMMAE are approved for the treatment of various hematologic malignancies, including relapsed or refractory Hodgkin lymphoma and systemic anaplastic large cell lymphoma. These ADCs have demonstrated efficacy in patients who have failed previous therapies, offering new treatment options for individuals with aggressive or resistant forms of these cancers. VcMMAE-based ADCs are also being investigated for the treatment of solid tumors, including breast cancer, lung cancer, and prostate cancer. Preclinical and clinical studies have shown promising results, with ADCs targeting tumor-associated antigens such as HER2 and Nectin-4 showing antitumor activity in a variety of solid tumor models. VcMMAE is being evaluated in combination with other anticancer agents, including chemotherapy drugs, targeted therapies, and immunotherapies, to enhance treatment efficacy and overcome resistance mechanisms in cancer cells. Combinations of VcMMAE-based ADCs with immune checkpoint inhibitors, for example, aim to leverage the cytotoxic effects of VcMMAE with the immunomodulatory properties of checkpoint blockade, leading to enhanced antitumor immune responses. References none |
| Market Analysis Reports |