Online Database of Chemicals from Around the World
| Epochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.epochem.com | |||
 | +86 (21) 6760-1595 6760-1597 | |||
 | +86 (21) 6760-1605 | |||
 | seth_wang@epochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Nanjing Chemfly Medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemfly.com | |||
 | +86 18910986335 | |||
 | info@chemfly.com | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Relybo Pharmachemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.relybopharma.com | |||
 | +86 (25) 8771-6566 | |||
 | susanchen@relybopharma.com | |||
| Chemical distributor since 2011 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Frontier Scientific Services, Inc. | USA | |||
|---|---|---|---|---|
 | www.frontierssi.com | |||
 | +1 (302) 266-6891 (888) 577-2734 | |||
 | +1 (302) 266-8296 | |||
 | customerservice@frontierssi.com | |||
| Chemical manufacturer | ||||
| Alfa Aesar. | USA | |||
|---|---|---|---|---|
 | www.alfa.com | |||
 | +1 (978) 521-6300 | |||
 | +1 (978) 521-6350 | |||
 | info@alfa.com | |||
| Chemical manufacturer | ||||
| Changzhou Wujin Minghuang Chemical Factory | China | |||
|---|---|---|---|---|
 | www.minghuangchem.com | |||
 | +86 (519) 8653-6539 +86 13606148116 | |||
 | +86 (519) 8652-8331 | |||
 | gxchem@jsmail.com.cn | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Chinese herbal medicine ingredients |
|---|---|
| Name | 2-Naphthaldehyde |
| Synonyms | 2-Naphthalenecarboxaldehyde |
| Molecular Structure | 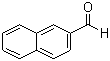 |
| Molecular Formula | C11H8O |
| Molecular Weight | 156.18 |
| CAS Registry Number | 66-99-9 |
| EC Number | 200-640-2 |
| SMILES | C1=CC=C2C=C(C=CC2=C1)C=O |
| Density | 1.2±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 58-61 °C (Expl.) |
| Index of Refraction | 1.676, Calc.* |
| Boiling Point | 299.5±9.0 °C (760 mmHg), Calc.*, 160 °C (19 torr) (Expl.) |
| Flash Point | 179.5±5.0 °C, Calc.* |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
2-Naphthaldehyde is an aromatic aldehyde with the molecular formula C(_{11})H(_8)O. Structurally, it consists of a naphthalene ring system with an aldehyde functional group located at the 2-position. This compound is notable for its aromatic stability and its ability to participate in a wide range of chemical reactions, making it a valuable intermediate in organic synthesis and materials science. The discovery and study of naphthaldehydes, including 2-naphthaldehyde, date back to the late 19th and early 20th centuries as chemists explored derivatives of naphthalene, a fundamental aromatic hydrocarbon. Early research focused on methods of functionalizing the naphthalene ring, particularly through oxidation and formylation techniques, to create compounds like 2-naphthaldehyde. Classical approaches, such as the Gattermann–Koch formylation, provided pathways to synthesize this compound. 2-Naphthaldehyde has gained attention for its reactivity, particularly in condensation reactions. It serves as a key precursor in the synthesis of a variety of heterocyclic compounds, dyes, and advanced organic materials. Its aldehyde group makes it highly reactive with nucleophiles, enabling the formation of Schiff bases, which are widely used in coordination chemistry and catalysis. In the dye industry, 2-naphthaldehyde is used in the production of azo dyes. Its aromatic framework and aldehyde functionality facilitate its coupling with amines and other intermediates to produce brightly colored pigments. These dyes find applications in textiles, printing, and coatings. Another notable application of 2-naphthaldehyde is in pharmaceuticals. It serves as a building block in the synthesis of biologically active molecules, including antifungal, antibacterial, and anticancer agents. Its aromatic structure and reactivity make it ideal for designing drug candidates and ligands for medicinal chemistry studies. 2-Naphthaldehyde also plays a role in materials science. Its derivatives are used in the design of fluorescent sensors and organic semiconductors. The conjugated system of the naphthalene ring imparts photophysical properties that are valuable for optoelectronic applications. Ongoing research on 2-naphthaldehyde explores its potential in advanced organic synthesis and green chemistry. Modified synthetic methods aim to improve yields, reduce environmental impact, and enhance its availability for industrial applications. The combination of its reactivity and versatility has made 2-naphthaldehyde an enduringly valuable compound in organic and materials chemistry, with applications that continue to expand in scope. References 2022. Novel allyl-hydrazones including 2,4-dinitrophenyl and 1,2,3-triazole moieties as optical sensor for ammonia and chromium ions in water. BMC Chemistry, 16(1). DOI: 10.1186/s13065-022-00820-2 1965. OXIDATIVE METABOLISM OF PHENANTHRENE AND ANTHRACENE BY SOIL PSEUDOMONADS. THE RING-FISSION MECHANISM. The Biochemical journal, 95(3). DOI: 10.1042/bj0950819 1960. Sensitive new test for aliphatic, aromatic, and heterocyclic aldehydes. Microchimica Acta, 48(4). DOI: 10.1007/bf01215431 |
| Market Analysis Reports |