Online Database of Chemicals from Around the World
| Shaoyang Kerey Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kery-pharm.com | |||
 | +86 (739) 231-2559 +86 13807397282 | |||
 | +86 (739) 231-2555 | |||
 | kereyinfo@126.com | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| HBCChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.hbcchem-inc.com | |||
 | +1 (510) 219-6317 | |||
 | +1 (510) 675-0318 | |||
 | sales@hbcchem-inc.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Handan Liye Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hdlyhk.com | |||
 | +86 (310) 816-6570 | |||
 | +86 (310) 816-6570 | |||
 | liye@hdlyhk.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Qingdao Xinnuo Pharmaceutical Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xinnuopharma.com | |||
 | +86 (531) 6656-1050 +86 18615688656 +86 18653156686 | |||
 | +86 (531) 6656-1050 | |||
 | sales@xinnuopharma.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hebei Brant Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.brantpharm.com | |||
 | +86 18713118368 | |||
 | lisa@brantpharm.com | |||
 | WhatsApp:0086 18713118368 | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Zibo Tiantangshan Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chinatts.com | |||
 | +86 (571) 745-5887 745-0218 | |||
 | +86 (571) 745-1321 | |||
 | manager@chinatts.com | |||
| Chemical manufacturer | ||||
| Jiangsu Tendenci Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tendencichem.com | |||
 | +86 (512) 5283-8508 | |||
 | +86 (512) 5283-8508 | |||
 | tjp@tendencichem.com | |||
| Chemical manufacturer since 1983 | ||||
| Xiamen Flint Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.flintchem.com | |||
 | +86 (592) 389-2837 | |||
 | +86 (592) 560-6787 | |||
 | sales@flintchem.com | |||
| Chemical distributor | ||||
| Classification | Chemical reagent >> Organic reagent >> Sulfonate / sulfinate |
|---|---|
| Name | Disodium 2,5-dihydro-5-thiooxo-1H-tetrazol-1-ylmethanesulfonate |
| Synonyms | 5-Mercapto-1H-tetrazole-1-methanesulfonic acid disodium salt |
| Molecular Structure | 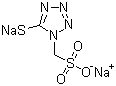 |
| Molecular Formula | C2H2N4Na2O3S2 |
| Molecular Weight | 240.17 |
| CAS Registry Number | 66242-82-8 |
| EC Number | 266-274-0 |
| SMILES | C(N1C(=NN=N1)[S-])S(=O)(=O)[O-].[Na+].[Na+] |
| Melting point | 247 $degree$C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
|
Disodium 2,5-dihydro-5-thiooxo-1H-tetrazol-1-ylmethanesulfonate is a water-soluble organosulfur compound featuring a tetrazole ring substituted with a thioxo group at the 5-position and a methanesulfonate group attached to the 1-position. Tetrazole derivatives are widely used in medicinal chemistry, coordination chemistry, and synthetic chemistry due to their high nitrogen content, acidity, and ability to act as bioisosteres of carboxylic acids. The presence of two sodium ions balances the sulfonate anion, enhancing solubility and stability in aqueous systems. Structurally, the molecule consists of a five-membered tetrazole ring with four nitrogen atoms, a hydrogen at the 2-position, a thioxo (C=S) group at the 5-position, and a methanesulfonate substituent at the 1-position. The sulfonate group is deprotonated and paired with two sodium cations, which increases the ionic character and facilitates dissolution in polar solvents. The tetrazole ring itself is electron-rich and can engage in hydrogen bonding, coordination to metals, and nucleophilic or electrophilic reactions, while the thioxo group adds nucleophilicity and potential for sulfur-centered chemistry. The synthesis of disodium 2,5-dihydro-5-thiooxo-1H-tetrazol-1-ylmethanesulfonate generally involves the reaction of a tetrazole precursor with a methanesulfonylating agent, followed by introduction of the thioxo functionality through sulfurization. Neutralization with sodium hydroxide or another suitable base generates the disodium salt. The reaction conditions are controlled to prevent decomposition of the tetrazole ring and to ensure selective functionalization at the 1- and 5-positions. The resulting disodium salt is stable, water-soluble, and suitable for use as a reagent or intermediate in further chemical transformations. In medicinal chemistry, tetrazole derivatives such as this compound are explored as bioisosteres of carboxylic acids due to similar acidity and hydrogen-bonding capacity, which can improve metabolic stability and membrane permeability. The thioxo group provides additional reactivity for interactions with nucleophilic residues in proteins, while the methanesulfonate functionality can enhance solubility and facilitate formulation in aqueous systems. This combination of features allows chemists to design compounds for enzyme inhibition, receptor modulation, or metal coordination. Beyond medicinal applications, this compound serves as a versatile intermediate in organic and inorganic chemistry. The tetrazole ring can undergo further derivatization, including alkylation, acylation, or cycloaddition reactions. The sulfonate group provides a handle for ionic interactions or coupling reactions, while the thioxo functionality allows for sulfur-based chemistry such as formation of thiol derivatives or coordination to metal centers. This makes the compound useful for synthesizing heterocyclic scaffolds, coordination complexes, or functional materials. Overall, disodium 2,5-dihydro-5-thiooxo-1H-tetrazol-1-ylmethanesulfonate is a multifunctional heterocyclic disodium salt combining a tetrazole ring, a thioxo group, and a sulfonate substituent. Its water solubility, nucleophilicity, and hydrogen-bonding potential make it a valuable intermediate for synthetic chemistry, medicinal chemistry, and the preparation of bioactive compounds. References 2024. Multifunctional hard tissue treating agent based on sulfhydryl and sulfonic acid group structural compound and application thereof. CN Patent. URL: CN-119015132-A 2024. Silver-copper alloy coating and preparation method thereof. CN Patent. URL: CN-118756270-A |
| Market Analysis Reports |