Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Georganics Ltd. | Slovakia | |||
|---|---|---|---|---|
 | www.georganics.sk | |||
 | +421 (33) 640-3132 | |||
 | +421 (33) 640-3133 | |||
 | georganics@georganics.sk | |||
| Chemical manufacturer since 1998 | ||||
| Ryan Scientific, Inc. | USA | |||
|---|---|---|---|---|
 | www.ryansci.com | |||
 | +1 (843)-884-4911 | |||
 | +1 (843) 884-5568 | |||
 | sales@ryansci.com | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Nitrile compound |
|---|---|
| Name | N-Cyanoacetylurethane |
| Synonyms | Ethyl N-(cyanoacetyl)carbamate; Ethyl cyanoacetylcarbamate; N-Carbethoxy-2-cyanoacetamide; NSC 59709 |
| Molecular Structure | 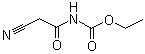 |
| Molecular Formula | C6H8N2O3 |
| Molecular Weight | 156.14 |
| CAS Registry Number | 6629-04-5 |
| EC Number | 229-615-4 |
| SMILES | CCOC(=O)NC(=O)CC#N |
| Density | 1.197±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 167-168 °C (Expl.) |
| Index of Refraction | 1.452, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
N-Cyanoacetylurethane is a versatile organic compound used primarily as an intermediate in various synthetic processes, especially within the pharmaceutical and agrochemical industries. It has a molecular formula of C5H6N2O3 and contains both cyano and urethane groups, which grant it specific reactivity, allowing it to participate in multiple synthetic reactions. The presence of the cyano group is particularly significant as it provides the compound with nucleophilic and electrophilic properties, making it adaptable to various chemical transformations. The discovery and synthesis of N-Cyanoacetylurethane can be traced to studies on urethane derivatives and cyano compounds, both known for their importance in medicinal and agricultural chemistry. Early studies highlighted the potential of cyanoacetyl compounds in constructing more complex molecules due to their functional versatility. By incorporating a urethane group, researchers found that N-Cyanoacetylurethane could serve as a stable and reactive intermediate in multi-step organic syntheses. Its ability to undergo reactions with amines, aldehydes, and ketones contributed to its early popularity among synthetic chemists aiming to build pharmacologically active compounds. One prominent application of N-Cyanoacetylurethane is in drug synthesis. The compound’s reactivity makes it suitable for the preparation of heterocyclic molecules, which are common scaffolds in medicinal chemistry. Heterocycles derived from N-Cyanoacetylurethane serve as core structures in various pharmaceuticals, such as antiviral and antibacterial agents. In these applications, N-Cyanoacetylurethane functions as a building block that enables the attachment of different functional groups, contributing to the development of structurally diverse therapeutic molecules. In agrochemical development, N-Cyanoacetylurethane is also valuable as a precursor in synthesizing compounds with pesticidal and herbicidal properties. The cyano and urethane functionalities in N-Cyanoacetylurethane facilitate its transformation into molecules that can act as inhibitors of specific biological pathways in pests or invasive plant species, thus aiding in crop protection and increasing agricultural productivity. N-Cyanoacetylurethane is additionally utilized in research settings to explore novel synthetic methodologies, particularly those aimed at developing efficient reaction pathways for cyanoacylation and urethane transformations. It is a popular choice in studies focusing on the design of new reaction conditions for mild and selective synthesis, where control over functional group compatibility is critical. Despite its many applications, handling N-Cyanoacetylurethane requires care, as cyano-containing compounds may exhibit toxicity if mishandled or improperly disposed of. Industrial and laboratory use of N-Cyanoacetylurethane is thus managed with safety protocols to minimize exposure risks and environmental impact. References 2018. 5-Amino-pyrazoles: potent reagents in organic and medicinal synthesis. Molecular Diversity, 22(4). DOI: 10.1007/s11030-018-9902-8 2010. Diversity-oriented, one-pot, multi-component synthesis of substituted uracil derivatives. Molecular Diversity, 14(4). DOI: 10.1007/s11030-010-9266-1 1968. N-(ω-carbalkoxyalkyl)ureas and their cyclic derivatives. III. N1-(ω-carbalkoxyalkyl)-5-cyanouracils based on ω-aminoalkanecarboxylic acids. Pharmaceutical Chemistry Journal, 2(1). DOI: 10.1007/bf00761090 |
| Market Analysis Reports |