Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Zison Pharmaceutical (shandong)co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zisonpharm.com | |||
 | +86 (0531) 8825-9693 +86 15069083822 | |||
 | tracy.li@zisonpharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2021 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Pure Bio Medical Jiangsu Ltd. | China | |||
|---|---|---|---|---|
 | www.purejs.cn | |||
 | +86 (025) 8560-7383 | |||
 | jiangziwei@purejs.cn | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | API >> Blood system medication >> Hemostatic drug |
|---|---|
| Name | Avatrombopag maleate |
| Synonyms | (Z)-but-2-enedioic acid;1-[3-chloro-5-[[4-(4-chlorothiophen-2-yl)-5-(4-cyclohexylpiperazin-1-yl)-1,3-thiazol-2-yl]carbamoyl]pyridin-2-yl]piperidine-4-carboxylic acid |
| Molecular Structure | 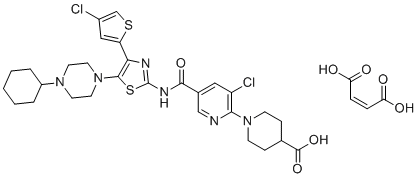 |
| Molecular Formula | C33H38Cl2N6O7S2 |
| Molecular Weight | 765.73 |
| CAS Registry Number | 677007-74-8 |
| SMILES | C1CCC(CC1)N2CCN(CC2)C3=C(N=C(S3)NC(=O)C4=CC(=C(N=C4)N5CCC(CC5)C(=O)O)Cl)C6=CC(=CS6)Cl.C(=CC(=O)O)C(=O)O |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P264-P270-P271-P280-P301+P312-P302+P352-P304+P340-P305+P351+P338-P312-P330-P362-P403+P233-P501 Details |
| SDS | Available |
|
Avatrombopag maleate is the maleate salt form of avatrombopag, an orally active small-molecule thrombopoietin receptor agonist used to increase platelet production in patients with thrombocytopenia. It is clinically used in adults with chronic liver disease undergoing procedures and in immune thrombocytopenia (ITP), depending on regulatory approvals in different regions. Avatrombopag was developed as part of efforts to create non-peptide, orally bioavailable agents that mimic the biological effects of thrombopoietin (TPO), a key endogenous cytokine that regulates platelet production. Instead of being a peptide like native thrombopoietin, avatrombopag is a synthetic small molecule designed to interact with the thrombopoietin receptor (c-Mpl) on megakaryocyte progenitor cells in the bone marrow. The mechanism of action involves agonism of the thrombopoietin receptor, leading to activation of intracellular signaling pathways such as JAK2/STAT5 and MAP kinase cascades. These pathways promote proliferation and differentiation of megakaryocytes, the bone marrow cells responsible for producing platelets. As a result, avatrombopag increases platelet counts in a dose-dependent manner. Structurally, avatrombopag is a heteroaromatic small molecule containing multiple substituted aromatic rings and heterocyclic components that allow it to bind selectively to the thrombopoietin receptor transmembrane domain. It does not bind to the same site as endogenous thrombopoietin but acts as a non-peptide receptor agonist that induces receptor activation through an alternative binding mechanism. The maleate salt form is used to improve the compound’s pharmaceutical properties, particularly solubility and stability in oral dosage formulations. Salt formation is a common strategy in drug development to optimize absorption and manufacturability. Avatrombopag was developed to address limitations of earlier thrombopoietin receptor agonists, including injectable peptide-based agents and other small molecules with dietary restrictions or drug interaction concerns. One of its advantages is that it does not require dietary restrictions for absorption, unlike some related agents that have reduced bioavailability when taken with food. After oral administration, avatrombopag is absorbed and distributed systemically, where it exerts its effect on bone marrow megakaryocyte precursors. It is metabolized primarily in the liver, and its pharmacokinetics allow for short-course dosing in specific clinical settings, such as pre-procedural platelet elevation in patients with chronic liver disease. Clinically, avatrombopag maleate is used to reduce the need for platelet transfusions in patients with thrombocytopenia undergoing medical or surgical procedures. In chronic immune thrombocytopenia, it helps increase and maintain platelet counts, reducing bleeding risk. Adverse effects associated with avatrombopag include headache, fatigue, and potential thrombotic events due to increased platelet counts. As with other thrombopoietin receptor agonists, monitoring is required to avoid excessive platelet elevation. Overall, avatrombopag maleate is an orally active thrombopoietin receptor agonist that stimulates megakaryocyte proliferation and platelet production through activation of c-Mpl signaling pathways. Its development represents an advance in the management of thrombocytopenia, providing a non-peptide, orally administered alternative to injectable or less convenient therapies. References 2018. Avatrombopag. Pharmaceutical Substances. URL: https://pharmaceutical-substances.thieme.com/ps/search-results?docUri=KD-16-0287 |
| Market Analysis Reports |