Online Database of Chemicals from Around the World
| Shanghai Rui Yun Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rychemical.com.cn | |||
 | +86 (21) 6726-7633 | |||
 | +86 (21) 6726-7655 | |||
 | sales@rychemical.com.cn | |||
 | WeChat: 13917251563 | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Premium supplier since 2009 | ||||
| Classification | Organic raw materials >> Organic sulfur compound |
|---|---|
| Name | Methyl 2,2-difluoro-2-(fluorosulfonyl)acetate |
| Molecular Structure | 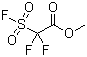 |
| Molecular Formula | C3H3F3O4S |
| Molecular Weight | 192.11 |
| CAS Registry Number | 680-15-9 |
| EC Number | 629-185-1 |
| SMILES | COC(=O)C(F)(F)S(=O)(=O)F |
| Density | 1.509 |
|---|---|
| Boiling point | 117-118 °C |
| Refractive index | 1.351 |
| Flash point | 47 °C |
| Hazard Symbols |   GHS02;GHS05 Danger Details GHS02;GHS05 Danger Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226-H314 Details | ||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P260-P264-P280-P301+P330+P331-P302+P361+P354-P303+P361+P353-P304+P340-P305+P354+P338-P316-P321-P363-P370+P378-P403+P235-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| Transport Information | UN 2920 | ||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Methyl 2,2-difluoro-2-(fluorosulfonyl)acetate is an organofluorine compound that has gained attention for its reactivity and application in organic synthesis. It features a methyl ester group attached to a difluorinated carbon, which is further bonded to a fluorosulfonyl group. This structural combination of fluorine and sulfonyl groups imparts the compound with unique chemical properties, making it a valuable reagent in various chemical transformations. The discovery of methyl 2,2-difluoro-2-(fluorosulfonyl)acetate emerged from research focused on the synthesis of fluorinated organic compounds. Fluorinated chemicals are widely used in the development of pharmaceuticals, agrochemicals, and advanced materials because of their stability, bioactivity, and ability to modify the behavior of organic molecules. The presence of both fluorine atoms and the reactive fluorosulfonyl group in this compound allows it to participate in diverse reactions, including nucleophilic substitution, addition reactions, and more specialized synthetic processes. One of the primary applications of methyl 2,2-difluoro-2-(fluorosulfonyl)acetate is in the preparation of fluorinated building blocks for drug development. Fluorinated esters such as this are particularly useful for modifying the pharmacological properties of molecules, improving their metabolic stability, bioavailability, and potency. The compound is often used to introduce fluorine atoms into molecules at key positions, which can significantly alter their interaction with biological targets. In addition to pharmaceutical applications, methyl 2,2-difluoro-2-(fluorosulfonyl)acetate plays a role in the synthesis of specialty chemicals, particularly those used in the manufacture of materials with enhanced durability and performance. Fluorinated materials are valued in industries such as electronics, aerospace, and energy storage due to their exceptional resistance to heat, chemicals, and mechanical stress. This compound serves as a starting material for producing these high-performance materials, where the combination of fluorine and sulfonyl functionality is crucial for achieving the desired properties. Ongoing research into fluorinated reagents like methyl 2,2-difluoro-2-(fluorosulfonyl)acetate continues to uncover new applications, particularly in the context of green chemistry. Its reactivity offers opportunities for more sustainable and efficient synthetic methods, contributing to the development of environmentally friendly technologies. References 2012. Methyl 2,2-Difluoro-2-(fluorosulfonyl)acetate, a Difluorocarbene Reagent with Reactivity Comparable to That of Trimethylsilyl 2,2-Difluoro-2-(fluorosulfonyl)acetate (TFDA). The Journal of Organic Chemistry, 77(10). DOI: 10.1021/jo300876z 2021. Contemporary synthetic strategies in organofluorine chemistry. Nature Reviews Methods Primers, 1(1). DOI: 10.1038/s43586-021-00042-1 2024. SuFEx Click Chemistry: Discovery to Applications. Materials Horizons: From Nature to Nanomaterials, 1(1). DOI: 10.1007/978-981-97-4596-8_8 |
| Market Analysis Reports |