Phenylsilane (C6H5SiH3) is an organosilicon compound consisting of a phenyl group directly bonded to a silicon atom, which in turn bears three hydrogens. It is a colorless, flammable liquid at room temperature and is notable for its utility as a reducing agent in organic synthesis. The Si–H bonds in phenylsilane are polarized and reactive, enabling hydride transfer to various substrates in catalytic and stoichiometric reactions.
Phenylsilane was first synthesized in the early 20th century during studies on organosilicon compounds, which were pioneered following the development of Grignard reagent chemistry. A common method for preparing phenylsilane involves the reaction of phenylmagnesium bromide with silicon tetrachloride, followed by reduction to yield the hydrosilane. Alternative methods include catalytic hydrogenolysis of phenylsilyl chlorides or redistribution reactions between phenylsilanes and other hydrosilanes.
In synthetic chemistry, phenylsilane is widely used as a mild and selective hydride source. It participates in hydrosilylation reactions with alkenes and alkynes, catalyzed by transition metal complexes such as platinum, rhodium, or nickel, to form organosilicon derivatives. It also serves as a stoichiometric reducing agent for the conversion of organic halides, carbonyl compounds, and imines to their reduced counterparts. In combination with metal catalysts, phenylsilane can reduce amides to amines under relatively mild conditions, a transformation valued in pharmaceutical synthesis.
Phenylsilane is also used in metal-catalyzed cross-coupling reactions, where it can act as a hydrogen donor in transfer hydrogenation processes. Its reactivity is influenced by the electron-withdrawing nature of the phenyl group, which modulates the Si–H bond polarity and facilitates selective activation in catalytic cycles. In organometallic chemistry, phenylsilane is a reagent for generating low-valent metal complexes through hydride transfer to metal centers.
In materials science, phenylsilane derivatives have been investigated as precursors for silicon-containing polymers and coatings. The phenyl substituent enhances thermal stability and imparts hydrophobicity, making phenylsilane-based materials attractive for protective and functional surface treatments.
Handling of phenylsilane requires care due to its flammability and potential to release hydrogen gas upon reaction with moisture or catalysts. It is typically stored under inert atmosphere to prevent degradation or unwanted reactions. Its relatively low toxicity compared to some organosilicon reagents makes it a practical choice in many laboratory-scale syntheses, though appropriate safety measures remain essential.
References
2024. Rare-earth metal bis(alkyl) amidopyridinate complexes in dehydrocoupling of anisole with hydrosilanes. Russian Chemical Bulletin, 73(10).
DOI: 10.1007/s11172-024-4404-6
1960. Orientation in the chlorination and bromination of phenylsilanes containing SiF3 and Si(CH3)3 groups. Bulletin of the Academy of Sciences of the USSR, Division of Chemical Science, 9(2).
DOI: 10.1007/bf00942894
1953. Grignard synthesis of tetra phenylsilane, tetra-p-tolysilane and fluorotri-p-tolylsilane. Bulletin of the Academy of Sciences of the USSR, Division of Chemical Science, 2(5).
DOI: 10.1007/bf01178854
|














































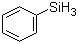

 GHS02;GHS07 Danger Details
GHS02;GHS07 Danger Details