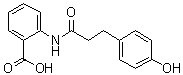
Hydroxyphenylpropionamide benzoic acid, also known as hydroxypropyl bishydroxyphenylpropionamide benzoic acid (HPA), is a compound obtained by the condensation reaction of propylamine and hydroxybenzoic acid. Its discovery can be traced back to research in the field of cosmetic chemistry, where scientists sought to develop new compounds with antioxidant and whitening benefits. HPA has been synthesized as a potential ingredient in skin care formulations for its ability to scavenge free radicals, inhibit tyrosinase activity, and reduce melanin synthesis in the skin. Its chemical structure consists of a propionamide benzoic acid moiety and a hydroxyphenyl group attached to the propylamine side chain.
Hydroxyphenylpropionamide benzoic acid is widely used in cosmetic formulations for its whitening and brightening properties. It inhibits the activity of melanin-producing tyrosinase, thereby reducing melanin formation in the skin. HPA has powerful antioxidant activity, making it an effective ingredient against oxidative stress and environmental damage to the skin. It scavenges free radicals generated by UV radiation, pollution and other external stressors, thereby preventing oxidative damage to cellular components and keeping skin healthy. Hydroxyphenylpropionamide benzoic acid also has anti-inflammatory properties that contribute to its healing effects on the skin. It inhibits the production of pro-inflammatory cytokines and mediators such as interleukin-1 (IL-1), tumor necrosis factor-alpha (TNF-alpha), and prostaglandins, thereby reducing inflammation and soothing irritated skin. Certain formulas containing hydroxyphenylpropionamidobenzoic acid may provide sun protection benefits by absorbing or reflecting UV radiation and preventing sunburn and UV-induced skin damage. Hydroxyphenylpropionamide benzoic acid has potential anti-aging effects on the skin by promoting collagen synthesis, improving skin firmness, and reducing the appearance of wrinkles and fine lines. It stimulates fibroblast activity, the cells responsible for the production of collagen and elastin fibers, thereby increasing skin elasticity and elasticity.
References
2017. Dihydroavenanthramide D inhibits mast cell degranulation and exhibits anti-inflammatory effects through the activation of neurokinin-1 receptor. Experimental Dermatology, 26(8).
DOI: 10.1111/exd.13265
2013. Dihydroavenanthramide D prevents UV-irradiated generation of reactive oxygen species and expression of matrix metalloproteinase-1 and -3 in human dermal fibroblasts. Experimental Dermatology, 22(11).
DOI: 10.1111/exd.12243
2009. Dermal and transdermal targeting of dihydroavenanthramide D using enhancer molecules and novel microemulsions. European journal of pharmaceutics and biopharmaceutics : official journal of Arbeitsgemeinschaft fur Pharmazeutische Verfahrenstechnik e.V, 72(3).
DOI: 10.1016/j.ejpb.2009.02.007
|
























 GHS07 Warning Details
GHS07 Warning Details