Online Database of Chemicals from Around the World
| Shandong Juntai Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.h2opharm.com | |||
 | +86 15106953682 | |||
 | sales01@h2opharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives >> Carboxylic esters and their derivatives |
|---|---|
| Name | 2,5-Dioxopyrrolidin-1-YL tetradecanoate |
| Synonyms | Succinimidyl myristate |
| Molecular Structure | 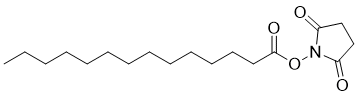 |
| Molecular Formula | C18H31NO4 |
| Molecular Weight | 325.44 |
| CAS Registry Number | 69888-86-4 |
| EC Number | 615-020-0 |
| SMILES | CCCCCCCCCCCCCC(=O)ON1C(=O)CCC1=O |
| Density | 1.1±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.489, Calc.* |
| Boiling Point | 420.2±28.0 °C (760 mmHg), Calc.* |
| Flash Point | 207.9±24.0 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319 Details | ||||||||||||
| Safety Statements | P264-P264+P265-P280-P302+P352-P305+P351+P338-P321-P332+P317-P337+P317-P362+P364 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
2,5-Dioxopyrrolidin-1-yl tetradecanoate, often referred to as an N-hydroxysuccinimide (NHS) ester of tetradecanoic acid, is a synthetic compound widely used in organic chemistry and biochemistry. Its structure consists of a long-chain fatty acid (tetradecanoic acid) esterified with an activated NHS group. This specific chemical combination renders the compound highly reactive, particularly useful in bioconjugation and peptide synthesis, where it facilitates the coupling of carboxyl groups to amines. The discovery of NHS esters traces back to efforts in the 20th century to develop better reagents for coupling reactions. NHS esters gained popularity due to their stability in comparison to other ester types and their ability to react specifically with primary amines in aqueous solutions. This precision and reactivity made NHS esters, such as 2,5-dioxopyrrolidin-1-yl tetradecanoate, ideal for modifying biomolecules in controlled chemical environments. In practical applications, 2,5-dioxopyrrolidin-1-yl tetradecanoate is widely used in the synthesis of lipid-like compounds and conjugation reactions. Its long tetradecanoic acid chain provides hydrophobic characteristics, making it useful in the development of amphiphilic molecules for use in lipid-based drug delivery systems. The NHS ester group reacts efficiently with primary amines, facilitating the formation of stable amide bonds, a key reaction in attaching small molecules, peptides, or other bioactive substances to carrier molecules or surfaces. In peptide synthesis, 2,5-dioxopyrrolidin-1-yl tetradecanoate serves as a coupling reagent to link fatty acid chains to peptides, a process known as lipidation. Lipidation is used to modify peptides to improve their membrane permeability, stability, and bioavailability. Peptides with lipid chains attached often exhibit enhanced pharmacokinetic properties, enabling better therapeutic efficacy, particularly in drug development targeting intracellular pathways. Furthermore, 2,5-dioxopyrrolidin-1-yl tetradecanoate is instrumental in surface chemistry. It is employed to immobilize proteins or peptides onto surfaces through covalent bonds, a technique commonly used in biosensor technologies and diagnostic assays. These applications capitalize on the compound’s ability to link biologically active molecules to solid supports without compromising their activity. This NHS ester derivative also finds utility in the synthesis of modified lipids for use in cosmetics and personal care products, where it serves as an intermediate for attaching functional groups to lipophilic compounds. Its role in creating emulsifying agents and enhancing the stability of formulations underscores its versatility in industrial applications. The broader use of NHS esters like 2,5-dioxopyrrolidin-1-yl tetradecanoate demonstrates the value of such reactive intermediates in enabling the synthesis of complex, functional molecules across biotechnology, pharmaceuticals, and industrial chemistry. References 1991. Labeling of adipocyte membranes by sulfo-N-succinimidyl derivatives of long-chain fatty acids: Inhibition of fatty acid transport. The Journal of Membrane Biology, 121(3). DOI: 10.1007/bf01951559 |
| Market Analysis Reports |