Online Database of Chemicals from Around the World
| Jiangsu Haixiang Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hx-chem.com | |||
 | +86 (519) 8019-1677 | |||
 | +86 (519) 8233-0677 | |||
 | michaelliorama@gmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| HBCChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.hbcchem-inc.com | |||
 | +1 (510) 219-6317 | |||
 | +1 (510) 675-0318 | |||
 | sales@hbcchem-inc.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Rivocean Group Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rivocean.com | |||
 | +86 (576) 8181-8908 | |||
 | +86 (576) 8181-8901 | |||
 | billior@rivocean.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Biosynth AG. | Switzerland | |||
|---|---|---|---|---|
 | www.biosynth.com | |||
 | +41 (71) 858-2020 | |||
 | +41 (71) 858-2030 | |||
 | welcome@biosynth.ch | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Changzhou Jintan ChengEn Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chengenchem.com | |||
 | +86 (519) 8019-0155 +86 13961176135 +86 13775159780 | |||
 | +86 (519) 8019-0155 | |||
 | sales@chengenchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Hangzhou Loop Biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.looppharm.com | |||
 | +86 (0571) 8526-0761 | |||
 | sales01@looppharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Hangzhou Zentra Bio-Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zentrachem.com | |||
 | 18321999949 | |||
 | steven@zentrachem.com | |||
 | QQ Chat | |||
 | WeChat: Hangzhou Zentra Bio-Chemical Co., Ltd. | |||
| Chemical distributor since 2025 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Changzhou Lianyi Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lianyichem.com | |||
 | +86 (519) 8613-1077 +86 13601508551 | |||
 | +86 (519) 8613-5266 | |||
 | sales@lianyichem.com | |||
| Chemical manufacturer since 1991 | ||||
| Molekula Ltd | UK | |||
|---|---|---|---|---|
 | www.molekula.com | |||
 | +44 (1747) 831-066 | |||
 | +44 (1747) 831-199 | |||
 | info@molekula.com | |||
| Chemical manufacturer | ||||
| Indofine Chemical Company, Inc. | USA | |||
|---|---|---|---|---|
 | www.indofinechemical.com | |||
 | +1 (888) 463-6346 | |||
 | +1 (908) 359-1179 | |||
 | info@indofinechemical.com | |||
| Chemical manufacturer since 1981 | ||||
| Tianmen Deyuan Chemical Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.deyuanchem.cn | |||
 | +86 (728) 535-3300 +86 13707211087 | |||
 | +86 (728) 535-3300 | |||
 | sales@deyuanchem.cn | |||
| Chemical manufacturer | ||||
| Cradlechem (Jiangsu) Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cradlechem.com | |||
 | +86 (519) 8571-5016 8503-3388 8571-6188 | |||
 | +86 (519) 8571-6288 | |||
 | cradlechem@163.com | |||
| Chemical manufacturer since 1988 | ||||
| Toronto Research Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.trc-canada.com | |||
 | +1 (416) 665-9696 | |||
 | +1 (416) 665-4439 | |||
 | info@trc-canada.com | |||
| Chemical manufacturer since 1982 | ||||
| Kingchem Inc. | USA | |||
|---|---|---|---|---|
 | www.kingchem.com | |||
 | +1 (800) 211-4330 (201) 825-9988 | |||
 | +1 (201) 825-9148 | |||
 | customer-service@kingchem.com | |||
| Chemical manufacturer since 1994 | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic ketone |
|---|---|
| Name | 2',6'-Dihydroxyacetophenone |
| Synonyms | 1-(2,6-Dihydroxyphenyl)ethan-1-one; 2-Acetyl-1,3-dihydroxybenzene; 2-Acetylresorcinol |
| Molecular Structure | 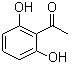 |
| Molecular Formula | C8H8O3 |
| Molecular Weight | 152.15 |
| CAS Registry Number | 699-83-2 |
| EC Number | 211-833-6 |
| SMILES | CC(=O)C1=C(C=CC=C1O)O |
| Density | 1.3±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 156 - 158 °C (Expl.) |
| Boiling point | 262.8±20.0 °C 760 mmHg (Calc.)* |
| Flash point | 127.0±18.3 °C (Calc.)* |
| Solubility | water: |
| Index of refraction | 1.595 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
2’,6’-Dihydroxyacetophenone is an aromatic ketone compound characterized by a benzene ring substituted with two hydroxyl groups at the 2’ and 6’ positions and an acetyl group (–COCH3) at the 1-position. The 2’ and 6’ positions are ortho to the acetyl substituent, creating a symmetrical dihydroxy substitution pattern on the aromatic ring, which significantly influences the compound’s chemical behavior and physical properties. The compound can be synthesized through selective hydroxylation of acetophenone derivatives or by chemical modification of suitable hydroxy-substituted aromatic precursors. Methods such as directed ortho-metalation or regioselective electrophilic substitution enable the introduction of hydroxyl groups at the desired positions. The synthesis requires precise control to achieve the correct substitution pattern without overreaction or undesired side products. Chemically, 2’,6’-dihydroxyacetophenone exhibits the typical reactivity of aromatic ketones and phenols. The hydroxyl groups act as hydrogen bond donors and participate in electrophilic aromatic substitution, oxidation, and condensation reactions. The acetyl group provides a reactive carbonyl site, capable of nucleophilic addition, enolization, and condensation, making the compound a useful intermediate in organic synthesis. The proximity of hydroxyl groups to the ketone facilitates intramolecular hydrogen bonding, which can affect reactivity and stability. Applications of 2’,6’-dihydroxyacetophenone include its use as an intermediate in the synthesis of pharmaceuticals, dyes, and bioactive natural products. Its structural features make it valuable for preparing complex heterocyclic compounds and coordination complexes with metal ions. The compound’s hydroxyl and ketone functionalities allow for diverse chemical transformations, broadening its applicability in medicinal chemistry and materials science. Analytical characterization employs nuclear magnetic resonance (¹H and ¹³C NMR) spectroscopy to confirm the positions of hydroxyl and acetyl substituents on the aromatic ring. Infrared (IR) spectroscopy detects characteristic absorption bands for hydroxyl groups near 3400 cm–1 and carbonyl groups near 1650–1700 cm–1. Mass spectrometry provides molecular weight verification and fragmentation consistent with the dihydroxyacetophenone structure. Physically, 2’,6’-dihydroxyacetophenone is typically obtained as a crystalline solid, exhibiting moderate solubility in polar organic solvents such as ethanol, methanol, and acetone. Its melting point depends on purity and crystalline form. The compound is stable under normal laboratory storage conditions but may undergo oxidation upon prolonged exposure to air and light. In summary, 2’,6’-dihydroxyacetophenone is a dihydroxy-substituted aromatic ketone with versatile chemical properties that facilitate its use as an intermediate in various synthetic applications. Its well-established synthesis, characterization, and reactivity profiles make it important in chemical research and industrial processes. References 2023. Novel cyclic undecapeptides immobilized on reduced graphene oxide surface for enhanced antibacterial properties. International Journal of Environmental Science and Technology, 20(6). DOI: 10.1007/s13762-023-05035-z 2021. Synthesis, antioxidant, in silico and computational investigation of 2,5-dihydroxyacetophenone derived chloro-substituted hydroxychalcones, hydroxyflavanones and hydroxyflavindogenides. Journal of Biomolecular Structure and Dynamics, 40(20). DOI: 10.1080/07391102.2021.1943527 2020. Reaction of 2-Acylphenols with α-Methylene Ketones. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-114-00188 |
| Market Analysis Reports |