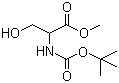
N-Boc-DL-serine methyl ester is an important compound in the field of organic chemistry, particularly within peptide synthesis. The compound consists of serine, an amino acid, which is protected by a Boc (tert-butyloxycarbonyl) group at its amine functional group and esterified at the carboxyl group. The Boc group is commonly used in synthetic chemistry to protect the amino group during reactions that involve other parts of the molecule. The methyl ester of serine allows the carboxyl group to be temporarily masked as well, further aiding in selective reactions. The “DL” designation indicates that the compound contains both the D- and L- stereoisomers of serine, making it racemic.
The discovery and widespread use of N-Boc-DL-serine methyl ester can be traced to advances in peptide synthesis and amino acid chemistry. The compound’s structural versatility makes it highly suitable for controlled reactions, where the presence of the protective Boc group and methyl ester enables chemists to conduct selective modifications without altering sensitive functional groups like the amino or carboxyl groups. This compound allows chemists to synthesize various peptide sequences or other complex molecules under mild conditions.
One of the major applications of N-Boc-DL-serine methyl ester is its use in peptide synthesis. Peptides, which are short chains of amino acids, are fundamental to many biological processes, serving roles as hormones, enzymes, and signaling molecules. During the synthesis of peptides, protecting groups like Boc are used to prevent unwanted side reactions that could occur at reactive functional groups, allowing for precise control over which part of the molecule is modified at any given step. After the desired chemical reaction occurs, the Boc group can be removed under acidic conditions, leaving the free amine intact.
In addition to its role in peptide synthesis, N-Boc-DL-serine methyl ester is used as an intermediate in the preparation of more complex molecules. It can serve as a building block for pharmaceuticals, where the serine residue is incorporated into drugs with specific biological targets. For example, serine-containing derivatives are known to be important in the synthesis of protease inhibitors, which are used to treat diseases such as HIV. The protective groups in N-Boc-DL-serine methyl ester enable the precise manipulation of molecular structures to create more stable and potent drugs.
Moreover, the methyl ester of serine allows for reactions at the carboxyl group without interference from the acidic properties of the free carboxyl group. This opens the door for the compound to be used in esterification reactions, which are crucial in both industrial and laboratory settings to produce esters from carboxylic acids.
In summary, N-Boc-DL-serine methyl ester plays a critical role in modern synthetic chemistry, particularly in the realm of peptide synthesis and pharmaceutical development. Its ability to protect both the amino and carboxyl functional groups allows for controlled and selective reactions, making it a versatile tool for chemists aiming to create complex organic molecules.
References
2011. Propylphosphonic Anhydride (T3P®)-Mediated One-Pot Rearrangement of Carboxylic Acids to Carbamates. Synthesis, 2011(6).
DOI: 10.1055/s-0030-1259964
|
























 GHS07 Warning Details
GHS07 Warning Details