Online Database of Chemicals from Around the World
| Earlier HK Company Limited | China | |||
|---|---|---|---|---|
 | www.earlierchem.com | |||
 | +86 (10) 6142-6769 +86 18616907430 | |||
 | +86 (10) 6142-6769 | |||
 | info@earlierchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Premium supplier since 2014 | ||||
| Classification | Organic raw materials >> Amino compound >> Acyclic monoamines, polyamines and their derivatives and salts |
|---|---|
| Name | Ethyldiisopropylamine |
| Synonyms | N,N-Diisopropylethylamine; N-Ethyldiisopropylamine; Huenig's base; DIEA; EDIA |
| Molecular Structure | 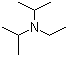 |
| Molecular Formula | C8H19N |
| Molecular Weight | 129.24 |
| CAS Registry Number | 7087-68-5 |
| EC Number | 230-392-0 |
| SMILES | CCN(C(C)C)C(C)C |
| Density | 0.782 |
|---|---|
| Melting point | -46 °C |
| Boiling point | 127 °C |
| Refractive index | 1.4123-1.4143 |
| Flash point | 6 °C |
| Water solubility | miscible |
| Hazard Symbols |     GHS02;GHS05;GHS06;GHS07 Danger Details GHS02;GHS05;GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H225-H302+H318+H331-H335 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P261-P264-P264+P265-P270-P271-P280-P301+P317-P303+P361+P353-P304+P340-P305+P354+P338-P316-P317-P319-P321-P330-P370+P378-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 1993; UN 2733 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Ethyldiisopropylamine, also known as Hunig's base or DIPEA (N,N-diisopropylethylamine), is an organic compound widely used in chemical synthesis. Its role as a non-nucleophilic base makes it indispensable in various fields such as pharmaceuticals, materials science, and organic chemistry. Ethyldiisopropylamine was first synthesized and identified in the mid-20th century, when chemists were trying to develop more efficient and selective bases for organic reactions. It has a chemical formula of C₈H₁₉N and is characterized by a bulky, branched structure. The structure includes an ethyl group attached to a nitrogen atom, which is also bonded to two isopropyl groups, resulting in significant steric hindrance around the nitrogen atom. This steric bulk makes DIPEA a non-nucleophilic base, meaning that it can deprotonate acids without participating in unwanted side reactions. It is typically a colorless to pale yellow liquid with a strong amine-like odor. It is soluble in many organic solvents, including dichloromethane and diethyl ether, but is not very soluble in water. DIPEA is widely used in peptide synthesis as a base to neutralize the acid formed during amino acid coupling. Its non-nucleophilic nature ensures that it does not interfere with the delicate formation of peptide bonds, making it a staple in protein and peptide synthesis. In the synthesis of amides and esters, DIPEA acts as a base, promoting the reaction between carboxylic acids (or their derivatives) and amines or alcohols. Its use ensures high yields and purity of the desired products by minimizing side reactions. DIPEA can be used as a catalyst in a variety of organic reactions, such as Michael additions, aldol condensations, and Heck couplings. Its effectiveness in these roles is attributed to its ability to deprotonate substrates and stabilize reaction intermediates without nucleophilic interference. In organic synthesis, protecting groups are often used to shield reactive sites on a molecule. DIPEA is commonly used for the deprotection of carbamate and ester groups, ensuring efficient removal without affecting other functional groups. The bulky structure of DIPEA prevents it from participating in nucleophilic side reactions, making it highly selective and effective in a variety of synthetic processes. Its solubility in organic solvents allows for easy incorporation into a variety of reaction conditions, enhancing its versatility. DIPEA is relatively stable under standard laboratory conditions and is easy to store and handle. Although DIPEA is a valuable reagent, it must be handled with care. It is corrosive and can cause severe irritation to the skin, eyes, and respiratory system. Appropriate safety measures include the use of personal protective equipment such as gloves, goggles, and working in a well-ventilated area. DIPEA should be stored in a cool, dry place away from acids and oxidizing agents. References 2013. Direct one-step labeling of cysteine residues on peptides with [(11)C]methyl triflate for the synthesis of PET radiopharmaceuticals. Amino Acids, 45(5). DOI: 10.1007/s00726-013-1562-5 2012. Parallel synthesis of 1,2,4-triazole derivatives using microwave and continuous-flow techniques. Molecular Diversity, 16(2). DOI: 10.1007/s11030-012-9357-2 2012. Click Chemistry with Polymers, Dendrimers, and Hydrogels for Drug Delivery. Pharmaceutical Research, 29(6). DOI: 10.1007/s11095-012-0683-y |
| Market Analysis Reports |