Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Boron Molecular Inc. | USA | |||
|---|---|---|---|---|
 | www.boronmolecular.com | |||
 | +1 (919) 981-4441 | |||
 | +1 (919) 981-4442 | |||
 | sales@boronmolecular-usa.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Boroncore LLC | China | |||
|---|---|---|---|---|
 | www.boroncore.com | |||
 | +86 (571) 8558-6753 | |||
 | custom@boroncore.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Zhejiang Free Trade Zone Violet Trading Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zjviolettech.com | |||
 | +86 (580) 811-8992 | |||
 | +86 (580) 811-9487 | |||
 | jenny@zjviolettech.com | |||
| Chemical distributor since 2019 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Combi-Blocks, Inc. | USA | |||
|---|---|---|---|---|
 | www.combi-blocks.com | |||
 | +1 (858) 635-8950 | |||
 | +1 (858) 635-8991 | |||
 | sales@combi-blocks.com | |||
| Chemical manufacturer | ||||
| AK Scientific, Inc | USA | |||
|---|---|---|---|---|
 | www.aksci.com | |||
 | +1 (510) 429-8835 | |||
 | +1 (510) 429-8836 | |||
 | sales@aksci.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic acid |
|---|---|
| Name | 3-Biphenylcarboxylic acid |
| Synonyms | 3-Phenylbenzoic acid |
| Molecular Structure | 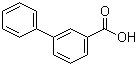 |
| Molecular Formula | C13H10O2 |
| Molecular Weight | 198.22 |
| CAS Registry Number | 716-76-7 |
| EC Number | 671-702-8 |
| SMILES | C1=CC=C(C=C1)C2=CC(=CC=C2)C(=O)O |
| Melting point | 164-169 °C |
|---|---|
| Water solubility | Insoluble |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H319-H335 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P304+P340-P305+P351+P338-P319-P330-P337+P317-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
3-Biphenylcarboxylic acid is an aromatic carboxylic acid characterized by a biphenyl structure with a carboxyl group (-COOH) attached to the third position of one of the phenyl rings. This compound is of significant interest due to its versatile chemical reactivity and its application in various fields, including organic synthesis, materials science, and medicinal chemistry. The discovery of 3-biphenylcarboxylic acid is part of the broader exploration of biphenyl derivatives, which have been studied extensively due to their stability and electronic properties. The introduction of a carboxyl group into the biphenyl structure adds a functional group that can participate in a wide range of chemical reactions, making 3-biphenylcarboxylic acid a valuable intermediate in organic synthesis. In organic synthesis, 3-biphenylcarboxylic acid is primarily used as a building block for the construction of more complex molecules. The carboxyl group allows for the formation of various derivatives through esterification, amidation, and other reactions. These derivatives are crucial in the synthesis of pharmaceuticals, agrochemicals, and advanced materials. For example, 3-biphenylcarboxylic acid can be used to create biphenyl-based ligands, which are important in catalytic processes and in the development of new materials with specific properties. One of the key applications of 3-biphenylcarboxylic acid is in the production of biphenyl-based polymers and materials. The biphenyl moiety provides rigidity and stability, which are desirable properties in the design of high-performance materials. These polymers can be used in a variety of applications, including electronic devices, sensors, and optical materials. The carboxyl group in 3-biphenylcarboxylic acid can participate in polymerization reactions, leading to the formation of high-molecular-weight materials with tailored properties. In medicinal chemistry, 3-biphenylcarboxylic acid is utilized as a precursor in the synthesis of various bioactive compounds. The biphenyl structure is a common motif in many pharmaceuticals, and the introduction of the carboxyl group allows for further functionalization and optimization of drug candidates. For instance, 3-biphenylcarboxylic acid derivatives have been explored for their potential as anti-inflammatory agents, enzyme inhibitors, and other therapeutic agents. The ability to modify the biphenyl structure with different functional groups makes 3-biphenylcarboxylic acid a versatile tool in drug discovery. Additionally, 3-biphenylcarboxylic acid finds applications in the synthesis of organic dyes and pigments. The biphenyl structure provides a platform for the development of colorants with specific absorption and emission properties. These dyes and pigments are used in various industries, including textiles, plastics, and coatings. The carboxyl group can be used to attach the dye molecules to different substrates, enhancing their performance and stability. The continued research into 3-biphenylcarboxylic acid highlights its importance in both fundamental and applied chemistry. Advances in synthetic methods and the development of new applications ensure that this compound remains a valuable asset in the design of new materials, pharmaceuticals, and industrial products. Its versatility and reactivity make it a key component in the ongoing exploration of biphenyl-based chemistry. References 2021. Synthesis of Benzo-Fused Cyclic Compounds via Rhodium-Catalyzed Decarboxylative Coupling of Aromatic Carboxylic Acids with Alkynes. Synthesis, 53(7). DOI: 10.1055/a-1416-6997 1996. Biphenyl-3-carboxylic acid at 296 and 203 K. Acta crystallographica. Section C, Crystal structure communications, 52(9). DOI: 10.1107/s0108270196003514 1989. New bacterial degradation of the biaryl ether structure. The Science of Nature, 76(11). DOI: 10.1007/bf00374127 |
| Market Analysis Reports |