Online Database of Chemicals from Around the World
| Beijing Huafeng United Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.huafenginfo.com | |||
 | +86 (10) 5786-2036 57862181 65405760 +86 13366977697 | |||
 | +86 (10) 5786-2181 | |||
 | huafenginfo@126.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Zhangjiagang Sunrise Fine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sfcc.com.cn | |||
 | +86 (574) 8797-0832 | |||
 | +86 (574) 8770-1738 | |||
 | info@sfcc.com.cn | |||
| Chemical manufacturer since 2000 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| 2A Pharmachem USA | USA | |||
|---|---|---|---|---|
 | www.2apharmachem.com | |||
 | +1 (630) 322-8887 | |||
 | +1 (630) 322-8885 | |||
 | sales@2abiotech.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Selleck Chemicals LLC | USA | |||
|---|---|---|---|---|
 | www.selleckchem.com | |||
 | +1 (713) 535-9129 | |||
 | +1 (832) 582-8590 | |||
 | info@selleckchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Shenzhen Happy Pharmaceuticals Ltd. | China | |||
|---|---|---|---|---|
 | www.happypharm.cn | |||
 | +86 (536) 737-6118 | |||
 | mark@happypharm.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Puho Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.puhopharma.com | |||
 | +86 13760689947 +86 (20) 6227-9976 | |||
 | sales@puhopharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Ningbo Easun Biological Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.easunbiotech.com | |||
 | +86 (574) 6208-9878 +86 18657120662 | |||
 | +86 (574) 6208-9877 | |||
 | sales@easunbiotech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| Sinova Corporation | USA | |||
|---|---|---|---|---|
 | www.sinovainc.com | |||
 | +1 (301) 961-1525 | |||
 | +1 (240) 235-4288 | |||
 | sales@sinovainc.com | |||
| Chemical manufacturer | ||||
| Classification | API >> Antineoplastic agents >> Antibiotic antineoplastic agents |
|---|---|
| Name | Pirarubicin |
| Synonyms | (7S,9S)-7-[(2R,4S,5S,6S)-4-amino-6-methyl-5-[(2R)-oxan-2-yl]oxyoxan-2-yl]oxy-6,9,11-trihydroxy-9-(2-hydroxyacetyl)-4-methoxy-8,10-dihydro-7H-tetracene-5,12-dione |
| Molecular Structure | 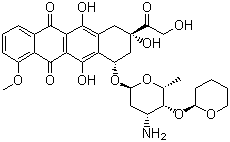 |
| Molecular Formula | C32H37NO12 |
| Molecular Weight | 627.64 |
| CAS Registry Number | 72496-41-4 |
| SMILES | C[C@H]1[C@H]([C@H](C[C@@H](O1)O[C@H]2C[C@@](CC3=C2C(=C4C(=C3O)C(=O)C5=C(C4=O)C(=CC=C5)OC)O)(C(=O)CO)O)N)O[C@@H]6CCCCO6 |
| Solubility | 100 mg/mL (DMSO) (Expl.) |
|---|---|
| Density | 1.5±0.1 g/cm3, Calc.* |
| Index of Refraction | 1.671, Calc.* |
| Boiling Point | 834.7±65.0 °C (760 mmHg), Calc.* |
| Flash Point | 458.6±34.3 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS08 Dander Details GHS08 Dander Details |
|---|---|
| Risk Statements | H340-H350-H360fd Details |
| Safety Statements | P203-P280-P318-P405-P501 Details |
| SDS | Available |
|
Pirarubicin is an anthracycline chemotherapy drug that has been used in the treatment of various cancers, particularly in the management of solid tumors and hematological malignancies. It was developed as a derivative of doxorubicin, one of the most well-known and widely used chemotherapy drugs, in an effort to improve its efficacy and reduce its cardiotoxicity. The drug was first synthesized in the 1980s and has since become an important treatment option, particularly in Japan and other parts of Asia, for cancers such as breast cancer, lung cancer, and leukemia. The discovery of pirarubicin involved modifying the structure of doxorubicin, a member of the anthracycline class of drugs known for their ability to intercalate into DNA, inhibit topoisomerase II, and generate free radicals. These actions result in the disruption of DNA replication and transcription, ultimately leading to cancer cell death. Pirarubicin differs from doxorubicin by the substitution of a hydroxyl group at the C-14 position of the aglycone part of the molecule. This modification reduces the drug's interaction with cellular membranes and may contribute to its reduced cardiotoxicity, one of the major side effects associated with doxorubicin use. Pirarubicin has proven to be particularly effective in the treatment of breast cancer, where it is often used as part of combination chemotherapy regimens. In these regimens, pirarubicin is typically combined with other chemotherapeutic agents to increase its efficacy and improve patient outcomes. It has also been used in the treatment of non-Hodgkin lymphoma, small cell lung cancer, and some types of leukemia, either as a single agent or as part of combination therapy. The key advantages of pirarubicin over its predecessor, doxorubicin, are its reduced cardiotoxicity and potentially lower incidence of side effects such as nausea, vomiting, and hair loss. Despite these advantages, pirarubicin still shares many of the same side effects as other anthracyclines, including myelosuppression (a reduction in bone marrow activity) and gastrointestinal toxicity. These side effects can be mitigated through careful dosing and supportive care, but they remain an important consideration in the management of patients receiving pirarubicin-based therapy. In recent years, research has focused on optimizing the use of pirarubicin by exploring different delivery methods and combination therapies. Studies have also investigated the potential of pirarubicin as part of targeted treatments for specific subtypes of cancer, with the goal of further improving its efficacy and minimizing its side effects. Additionally, the development of liposomal formulations of pirarubicin is being explored as a way to increase its targeted delivery to tumor sites, potentially reducing systemic exposure and toxicity. In conclusion, pirarubicin remains an important chemotherapy drug in the treatment of various cancers, offering a promising alternative to doxorubicin with reduced cardiotoxicity. While its side effects can be significant, its effectiveness, particularly in combination therapies, continues to make it a valuable tool in the fight against cancer. References 2023. A preclinical secondary pharmacology resource illuminates target-adverse drug reaction associations of marketed drugs. Nature Communications, 14(1). DOI: 10.1038/s41467-023-40064-9 2024. Adenylate cyclase 1 knockdown attenuates pirarubicin-induced cardiotoxicity. Clinical and Experimental Pharmacology and Physiology, 51(9). DOI: 10.1111/1440-1681.13920 2024. Pirarubicin combined with TLR3 or TLR4 agonists enhances anti-tumor efficiency. International Immunopharmacology, 142. DOI: 10.1016/j.intimp.2024.113068 |
| Market Analysis Reports |