Online Database of Chemicals from Around the World
| Changzhou Renke Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.renkechem.com | |||
 | +86 (519) 8685-7039 8371-7121 +86 13775109918 | |||
 | +86 (519) 8685-7039 | |||
 | rjw@renkechem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shandong Huali Bio-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hualibiochem.com | |||
 | +86 (533) 790-2030 +86 15601620667 +86 13611789960 | |||
 | +86 (533) 790-2030 / (21) 5756-8845 | |||
 | sdwhali@163.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Intatrade Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.intatrade.de | |||
 | +49 (3493) 605-465 | |||
 | +49 (3493) 605-470 | |||
 | sales@intatrade.de | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Enki Biopharmaceuticals (Shanghai) Limited | China | |||
|---|---|---|---|---|
 | www.enkibiopharma.com | |||
 | +86 (21) 5768-0965 +86 13916707528 | |||
 | +86 (21) 5768-0922 | |||
 | info@enkibiopharma.com | |||
 | QQ Chat | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Molcore Biopharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.molcore.com | |||
 | +86 (571) 8102-5280 | |||
 | sales@molcore.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Wonderchem Co., Limited | Hong Kong | |||
|---|---|---|---|---|
 | www.wonder-chem.com | |||
 | +852 8192-5218 | |||
 | sales@wonder-chem.com | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Marshallton Research Laboratories | USA | |||
|---|---|---|---|---|
 | www.marshalltonlabs.com | |||
 | +1 (336) 983-2131 | |||
 | +1 (336) 983-0096 | |||
 | marshallton@windstream.net | |||
| Chemical manufacturer since 1974 | ||||
| Melford Laboratories Limited | UK | |||
|---|---|---|---|---|
 | www.melford.co.uk | |||
 | +44 (1449) 741-178 | |||
 | +44 (1449) 741-217 | |||
 | pfranklin@melford.co.uk sales@melford.co.uk | |||
| Chemical manufacturer since 1985 | ||||
| Matrix Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.matrixscientific.com | |||
 | +1 (803) 788-9494 | |||
 | +1 (803) 788-9419 | |||
 | sales@matrixscientific.com | |||
| Chemical manufacturer | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Ricci Chimica | Italy | |||
|---|---|---|---|---|
 | www.riccichimica.com | |||
 | +39 (75) 692-9241 | |||
 | +39 (75) 592-6595 | |||
 | info@riccichimica.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| AOBChem USA | USA | |||
|---|---|---|---|---|
 | www.aobchem.com | |||
 | +1 (323) 382-7678 | |||
 | +1 (213) 687-8698 | |||
 | sales@aobchem.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic acid |
|---|---|
| Name | 3,5-Bis(trifluoromethyl)benzoic acid |
| Synonyms | 3,5-Di(trifluoromethyl)benzoic acid |
| Molecular Structure | 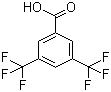 |
| Molecular Formula | C9H4F6O2 |
| Molecular Weight | 258.12 |
| CAS Registry Number | 725-89-3 |
| EC Number | 211-970-1 |
| SMILES | C1=C(C=C(C=C1C(F)(F)F)C(F)(F)F)C(=O)O |
| Density | 1.42 g/mL |
|---|---|
| Melting point | 140-144 °C |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
3,5-Bis(trifluoromethyl)benzoic acid was discovered through organic synthesis research focused on developing fluorinated organic compounds with unique properties. Chemists aimed to introduce multiple trifluoromethyl groups into a benzoic acid framework to enhance its reactivity, stability, and physicochemical properties. Through synthetic routes involving multi-step fluorination reactions, 3,5-Bis(trifluoromethyl)benzoic acid was successfully synthesized and characterized. Its discovery represented a significant advancement in fluorine chemistry, providing a new building block for the synthesis of fluorinated aromatic compounds with potential applications in pharmaceuticals, agrochemicals, materials science, and organic synthesis. 3,5-Bis(trifluoromethyl)benzoic acid serves as a versatile intermediate in the synthesis of pharmaceutical compounds, including fluorinated drugs and bioactive molecules. Its trifluoromethyl groups impart unique physicochemical properties, such as increased lipophilicity, metabolic stability, and protein binding affinity, which are advantageous for drug candidates. Derivatives of 3,5-Bis(trifluoromethyl)benzoic acid are valuable building blocks for the synthesis of fluorinated heterocycles and drug-like scaffolds. These compounds are utilized by medicinal chemists to design and optimize drug candidates with enhanced potency, selectivity, and pharmacokinetic properties. 3,5-Bis(trifluoromethyl)benzoic acid derivatives are employed in the synthesis of fluorinated pesticides and herbicides. These compounds exhibit improved efficacy, resistance to environmental degradation, and reduced ecological impact compared to their non-fluorinated counterparts, making them valuable components of modern agricultural chemical formulations. 3,5-Bis(trifluoromethyl)benzoic acid derivatives are utilized in the synthesis of fluorinated polymers with enhanced properties for specific applications. These polymers exhibit improved thermal stability, chemical resistance, and surface hydrophobicity, making them suitable for coatings, membranes, and electronic materials. 3,5-Bis(trifluoromethyl)benzoic acid serves as a precursor for the synthesis of fluorine-containing reagents and organofluorine compounds. Its trifluoromethyl groups enable the introduction of fluorine atoms into organic molecules through various synthetic transformations, including nucleophilic substitution, metal-catalyzed fluorination, and electrophilic fluorination reactions. Derivatives of 3,5-Bis(trifluoromethyl)benzoic acid are utilized as fluorine NMR probes in chemical and biological research. Their unique chemical shifts and sensitivity to environmental changes make them valuable tools for studying molecular structure, dynamics, and interactions in complex systems. References 2021. Milled Dry Ice as a C1 Source for the Carboxylation of Aryl Halides. Synlett : accounts and rapid communications in synthetic organic chemistry, 32(10). DOI: 10.1055/a-1384-0159 2019. Aqueous Flow Hydroxycarbonylation of Aryl Halides Catalyzed by an Amphiphilic Polymer-Supported Palladium-Diphenylphosphine Catalyst. Synlett, 30(8). DOI: 10.1055/s-0037-1611769 2002. Application of (1)H- and (19)F-NMR spectroscopy in the investigation of the urinary and biliary excretion of 3,5-, 2,4-ditrifluoromethylbenzoic and pentafluorobenzoic acids in rat. Xenobiotica; the fate of foreign compounds in biological systems, 32(1). DOI: 10.1080/00498250210131590 |
| Market Analysis Reports |