Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Shanghai RC Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rcc.net.cn | |||
 | +86 (21) 5032-2175 5866-1250 ex 603 | |||
 | +86 (21) 5032-2176 / 5866-1251 | |||
 | sales@rcc.net.cn chad@rcc.net.cn | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Carbone Scientific Co., Ltd. | UK | |||
|---|---|---|---|---|
 | www.carbonesci.com | |||
 | +44 (870) 486-8629 | |||
 | +44 (870) 288-7399 | |||
 | sales@carbonesci.com | |||
| Chemical distributor | ||||
| Shineme Chemfar Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemfar.com | |||
 | +86 (731) 8471-8061 | |||
 | +86 (731) 8471-6269 | |||
 | sales@chemfar.com | |||
| Chemical manufacturer since 1996 | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives >> Carboxylic esters and their derivatives |
|---|---|
| Name | Dimethyl 3-hydroxyglutarate |
| Synonyms | 3-Hydroxyglutaric acid dimethyl ester |
| Molecular Structure | 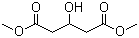 |
| Molecular Formula | C7H12O5 |
| Molecular Weight | 176.17 |
| CAS Registry Number | 7250-55-7 |
| SMILES | COC(=O)CC(CC(=O)OC)O |
| Density | 1.2±0.1 g/cm3, Calc.*, 1.192 g/mL (Expl.) |
|---|---|
| Index of Refraction | 1.444, Calc.*, 1.442 (Expl.) |
| Boiling Point | 265.6±20.0 °C (760 mmHg), Calc.*, 313.2-314.6 °C (Expl.) |
| Flash Point | 103.4±15.3 °C, Calc.*, 113 °C (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
Dimethyl 3-hydroxyglutarate is an organic compound and ester derivative of 3-hydroxyglutaric acid, characterized by the presence of two methyl ester groups and a central hydroxyl functionality. It is a colorless to pale yellow liquid under ambient conditions and exhibits good solubility in organic solvents such as methanol, dichloromethane, and ethanol. The molecular formula of dimethyl 3-hydroxyglutarate is C7H12O5, and its structure features a linear five-carbon chain with functional groups positioned to allow further derivatization, making it a versatile intermediate in organic synthesis. The compound is not known to occur naturally and is typically synthesized via esterification of 3-hydroxyglutaric acid with methanol in the presence of acid catalysts. Alternative routes involve the selective hydroxylation of dimethyl glutarate, allowing introduction of the hydroxyl group at the C-3 position under controlled conditions. Dimethyl 3-hydroxyglutarate has found application in multiple areas of research and industry. Its structure is useful in the preparation of fine chemicals and pharmaceuticals, where the hydroxyl and ester groups serve as functional handles for further transformations. One of the significant uses of this compound is in the synthesis of chiral intermediates for pharmaceutical compounds, particularly where enantiomerically pure diols or amino alcohols are desired. In synthetic organic chemistry, it has been used as a precursor to β-hydroxy ketones and related compounds via selective oxidation or alkylation reactions. Moreover, the compound’s reactivity toward nucleophilic substitution and ester cleavage makes it an attractive intermediate for the development of heterocyclic scaffolds and biologically active molecules. Studies have also explored its use in biocatalysis, where enzyme-catalyzed transformations of dimethyl 3-hydroxyglutarate have been investigated to yield optically pure hydroxy acids or related compounds. These transformations are valuable for green chemistry approaches and sustainable synthesis methods. References 1999. Sensitivity and specificity of free and total glutaric acid and 3-hydroxyglutaric acid measurements by stable-isotope dilution assays for the diagnosis of glutaric aciduria type I. Journal of Inherited Metabolic Disease, 22(8). DOI: 10.1023/a:1005683222187 2007. Use of Lipases in Organic Synthesis. Industrial Enzymes. DOI: 10.1007/1-4020-5377-0_18 2017. Catalytic Enantioselective Desymmetrization of Meso Compounds in Total Synthesis of Natural Products: Towards an Economy of Chiral Reagents. Synthesis, 49(18). DOI: 10.1055/s-0036-1589493 |
| Market Analysis Reports |