Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Shanghai RC Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rcc.net.cn | |||
 | +86 (21) 5032-2175 5866-1250 ex 603 | |||
 | +86 (21) 5032-2176 / 5866-1251 | |||
 | sales@rcc.net.cn chad@rcc.net.cn | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Tyger Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.tygersci.com | |||
 | +1 (609) 434-0144 | |||
 | +1 (609) 434-0143 | |||
 | sales@tygersci.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Werlchem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.werlchem.com | |||
 | +86 (23) 6752-1957 | |||
 | +86 (23) 6751-1575 | |||
 | sales@werlchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| SL Drugs and Pharmaceuticals Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.sldrugs.com | |||
 | +91 (40) 6661-1133 | |||
 | +91 (40) 2375-1130 | |||
 | enquiry@sldrugs.com | |||
| Chemical distributor since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| 2A Pharmachem USA | USA | |||
|---|---|---|---|---|
 | www.2apharmachem.com | |||
 | +1 (630) 322-8887 | |||
 | +1 (630) 322-8885 | |||
 | sales@2abiotech.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Porphyrin Systems | Germany | |||
|---|---|---|---|---|
 | www.porphyrin-systems.de | |||
 | +49 (40) 6466-5860 | |||
 | +49 (40) 6466-5861 | |||
 | hombrecher@porphyrin-systems.de | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Amatek Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amtchem.com | |||
 | +86 (512) 5835-6800 5858-4128 | |||
 | +86 (512) 5858-4118 | |||
 | amtchimie@gmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Witofly Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.witofly.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales@witofly.com | |||
 | QQ Chat | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Keminntek Laboratories | India | |||
|---|---|---|---|---|
 | www.keminnteklabs.com | |||
 | +91 (951) 512-8135 | |||
 | sales@keminnteklabs.com | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Shanghai Tajilin Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tajilin.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales003@tajilin.com | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Sichuan Hengkang Science And Technology Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hengkangpharma.com | |||
 | +86 (028) 8862-8155 | |||
 | hk999@hengkangtech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Atlantic Research Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.atlantic-chemicals.com | |||
 | +44 (8707) 746-454 | |||
 | +44 (8707) 746-455 | |||
 | info@atlantic-chemicals.com | |||
| Chemical manufacturer | ||||
| Ryan Scientific, Inc. | USA | |||
|---|---|---|---|---|
 | www.ryansci.com | |||
 | +1 (843)-884-4911 | |||
 | +1 (843) 884-5568 | |||
 | sales@ryansci.com | |||
| Chemical manufacturer | ||||
| Apollo Scientific Ltd. | UK | |||
|---|---|---|---|---|
 | www.apolloscientific.co.uk | |||
 | +44 (161) 406-0505 | |||
 | +44 (161) 406-0506 | |||
 | sales@apolloscientific.co.uk | |||
| Chemical manufacturer | ||||
| Chess GmbH | Germany | |||
|---|---|---|---|---|
 | www.chess-chem.com | |||
 | +49 (621) 318-9794 | |||
 | +49 (621) 318-9691 | |||
 | sales@chess-chem.com | |||
| Chemical manufacturer | ||||
| Syntech Labs, Inc. | USA | |||
|---|---|---|---|---|
 | www.syntechlabs.com | |||
 | +1 (732) 545-8380 | |||
 | +1 (732) 545-6881 | |||
 | info@syntechlabs.com | |||
| Chemical manufacturer | ||||
| CNH Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.cnhtechnologies.com | |||
 | +1 (781) 933-0362 | |||
 | +1 (781) 933-1839 | |||
 | info@cnhtechnologies.com | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Wako Pure Chemical Industries, Ltd. | Japan | |||
|---|---|---|---|---|
 | www.wako-chem.co.jp | |||
 | +81 (6) 6203-3741 | |||
 | +81 (6) 6201-5964 | |||
 | wkhk.info@fujifilm.com | |||
| Chemical manufacturer since 1922 | ||||
| Aaron Chemistry GmbH | Germany | |||
|---|---|---|---|---|
 | www.aaron-chemistry.de | |||
 | +49 (8823) 917-521 | |||
 | +49 (8823) 917-523 | |||
 | sales@aaron-chemistry.de | |||
| Chemical manufacturer | ||||
| Syntastic AB | Sweden | |||
|---|---|---|---|---|
 | www.syntastic.se | |||
 | +46 (70) 592-7309 | |||
 | +46 (8) 665-0773 | |||
 | sales@syntastic.se | |||
| Chemical manufacturer | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridine compound >> Pyridine derivative |
|---|---|
| Name | 4-N-BOC-Aminopiperidine |
| Synonyms | 4-N-(tert-Butoxycarbonyl)aminopiperidine |
| Molecular Structure | 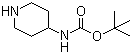 |
| Molecular Formula | C10H20N2O2 |
| Molecular Weight | 200.28 |
| CAS Registry Number | 73874-95-0 |
| EC Number | 616-026-6 |
| SMILES | CC(C)(C)OC(=O)NC1CCNCC1 |
| Density | 1.0±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 162 - 166 °C (Expl.) |
| Boiling point | 304.8±31.0 °C 760 mmHg (Calc.)* |
| Flash point | 138.2±24.8 °C (Calc.)* |
| Index of refraction | 1.48 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
4-(N-BOC-amino)piperidine is a protected amino piperidine derivative featuring a tert-butoxycarbonyl (BOC) group on the nitrogen atom of the piperidine ring, with an amine substituent at the 4-position. The molecular formula is C10H20N2O2, and the compound typically appears as a white crystalline solid. It is soluble in common organic solvents such as methanol, ethanol, dichloromethane, and ethyl acetate. The BOC protecting group enhances stability and allows selective deprotection under mild acidic conditions, making this compound a versatile building block in organic synthesis. In organic chemistry, 4-(N-BOC-amino)piperidine is widely used in pharmaceutical intermediate synthesis, particularly in the design of aminopiperidine scaffolds. Removal of the BOC group under acidic treatment or hydrogenolytic conditions liberates the free amine, which can then undergo further functionalization. The 4-amino functionality enables conjugation to pharmacophores or the creation of heterocyclic structures via reductive amination and coupling chemistry. As a result, the molecule serves as a key entry point for constructing complex, biologically active compounds. Synthetic routes to this compound commonly begin with piperidin-4-one derivatives. One method involves reacting N-benzyl-4-piperidone to introduce the BOC functionality, followed by methylenation to install functionality at the 4-position, and finally, catalytic hydrogenation to remove protecting groups and yield the desired BOC-protected amino compound in high yield. Alternative synthetic strategies employ direct BOC protection of 4-aminopiperidine or multistep transformations involving ketal intermediates to streamline the reaction sequence. 4-(N-BOC-amino)piperidine has found application in medicinal chemistry for elaborating libraries of aminopiperidine derivatives. These derivatives are investigated as candidates targeting muscarinic receptors, central nervous system pathways, viral entry receptors such as CCR5, and enzyme inhibition targets. For example, in DPP-4 inhibitor research, aminopiperidine analogues, which can be derived from BOC-protected precursors, are valuable for designing antidiabetic compounds with tailored receptor interactions. Commercially, this compound is offered by chemical suppliers in high-purity grades (typically ≥96–98 %), often with a melting point of approximately 162–166 °C. It is stored under refrigerated or ambient conditions and handled using standard protective equipment to guard against irritation. The compound is not considered particularly volatile but should be managed carefully in laboratory settings. In summary, 4-(N-BOC-amino)piperidine is a strategically protected aminopiperidine intermediate with broad utility in organic and medicinal chemistry. Its structural features—BOC-protected nitrogen and a reactive amino substituent—provide flexibility for downstream functionalization, making it a valuable tool in complex molecule synthesis. References 2025. Discovery, optimization and biological evaluation of chromone derivatives as novel BRD4 inhibitors. Medicinal Chemistry Research, 34(3). DOI: 10.1007/s00044-025-03380-x 2023. Discovery of pyrrolo[2,1-f][1,2,4]triazine-based inhibitors of adaptor protein 2-associated kinase 1 for the treatment of pain. Medicinal Chemistry Research, 32(6). DOI: 10.1007/s00044-023-03079-x 2022. Exploration of NMI-MsCl mediated amide bond formation for the synthesis of novel 3,5-substituted-1,2,4-oxadiazole derivatives: synthesis, evaluation of anti-inflammatory activity and molecular docking studies. Molecular Diversity, 27(5). DOI: 10.1007/s11030-022-10536-z |
| Market Analysis Reports |