Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Water Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.water-chemical.com | |||
 | +86 (21) 6856-1349 | |||
 | +86 (21) 5156-1796 | |||
 | info@water-chemical.com | |||
| Chemical manufacturer since 1994 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Sinocure Chemical Group | China | |||
|---|---|---|---|---|
 | www.sinocurechem.com | |||
 | +86 15550440621 | |||
 | info@sinocurechem.com | |||
| Chemical manufacturer since 2020 | ||||
| Celanese Chemicals | USA | |||
|---|---|---|---|---|
 | www.celanese.com | |||
 | +1 (972) 443-4000 | |||
 | +1 (972) 443-2060 | |||
 | info@celanese.com | |||
| Chemical manufacturer | ||||
| Shanghai Xiechen Trading & Developing Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xiechen.com | |||
 | +86 (21) 5169-1169 | |||
 | +86 (21) 5290-8698 | |||
 | 2691179155@qq.com | |||
| Chemical distributor | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives >> Carboxylic esters and their derivatives |
|---|---|
| Name | Butyl glycolate |
| Synonyms | Butyl hydroxyacetate |
| Molecular Structure | 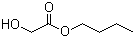 |
| Molecular Formula | C6H12O3 |
| Molecular Weight | 132.16 |
| CAS Registry Number | 7397-62-8 |
| EC Number | 230-991-7 |
| SMILES | CCCCOC(=O)CO |
| Density | 1.019 |
|---|---|
| Boiling point | 187-190 °C |
| Refractive index | 1.427 |
| Flash point | 74 °C |
| Hazard Symbols |  GHS05 Danger Details GHS05 Danger Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H318 Details | ||||||||||||||||||||
| Safety Statements | P264+P265-P280-P305+P354+P338-P317 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Butyl glycolate, a chemical compound with the formula C₆H₁₂O₄, is an ester of glycolic acid and butanol. It is known for its role in various industrial applications due to its unique chemical properties, which include solubility in both water and organic solvents. The substance is particularly valuable in formulations requiring a balance between solubility and reactivity. The discovery of butyl glycolate dates back to the early 20th century when chemists began exploring esters as intermediates and solvents in chemical reactions. Its synthesis typically involves the esterification of glycolic acid with butanol under acidic conditions. This process produces a compound with useful characteristics, such as a low vapor pressure and moderate polarity, making it suitable for a range of applications. In industry, butyl glycolate is widely utilized as a solvent in coatings and paint formulations. Its excellent solvency properties allow it to dissolve a variety of resins, pigments, and other components, which contributes to the formulation of high-quality paints with desirable properties such as smooth application, even drying, and enhanced adhesion. Additionally, its ability to evaporate slowly helps to prevent issues like pinholing and orange peel in finished coatings. Beyond its use in coatings, butyl glycolate is also employed as a solvent in the manufacturing of adhesives and sealants. It helps in achieving the desired consistency and spreading characteristics of these products, ensuring strong adhesion and durability. The substance's solvent properties are beneficial in formulations that require a balance between viscosity and evaporation rate, which is crucial for achieving optimal performance in various adhesive applications. In the fragrance industry, butyl glycolate is used as a carrier and solvent for perfumes and aromatic compounds. Its mild odor and compatibility with various fragrance ingredients make it an ideal choice for creating stable and well-balanced fragrance formulations. The compound helps in blending different fragrance notes and ensuring uniformity in the final product. Furthermore, butyl glycolate is explored for use in the pharmaceutical industry as a solvent for drug formulations. Its ability to dissolve a wide range of active pharmaceutical ingredients and its relatively low toxicity profile make it a candidate for developing oral and topical pharmaceutical products. Overall, butyl glycolate's versatility and effectiveness as a solvent and carrier in multiple industries highlight its importance in both traditional and innovative applications. Its role in enhancing product performance and stability continues to be an area of active research and development. References 1964. Synthesis of glyoxal and glycolic acid. Bulletin of the Academy of Sciences of the USSR, Division of chemical science. DOI: 10.1007/bf00845311 1989. Antimicrobial properties of some erucic acid-glycolic acid derivatives. Journal of the American Oil Chemists' Society, 66(7). DOI: 10.1007/bf02682611 |
| Market Analysis Reports |